Lessons From Europe’s Loss of Biopharma Leadership, and Its Attempts to Recover
Europe once led the world in biopharmaceutical innovation, but it lost ground after adopting policies that weakened incentives for R&D and innovation. America must learn from Europe’s experience to preserve its own biopharma leadership and the related economic benefits and access to the most innovative drugs.
KEY TAKEAWAYS
Key Takeaways
Contents
Europe’s Biopharmaceuticals Decline: A Cautionary Tale. 3
U.S. Biopharmaceutical Leadership and Its Foundations 8
Current U.S. Policy Missteps 11
Constructive Policy Alternatives 15
Europe’s Efforts to Reclaim Biopharmaceutical Leadership. 16
Introduction
Over the past several decades, global leadership in biopharmaceutical innovation has shifted dramatically. Once the undisputed center of drug discovery and development, Europe gradually fell behind as policy choices weakened incentives for investment, slowed progress, and constrained industry growth. During the same period, the United States built a policy environment far more conducive to innovation—one that supported research, rewarded risk taking, and attracted scientists, capital, and leading firms from around the world. As Europe’s policy environment became less attractive for high-risk biopharmaceutical research and development (R&D), firms increasingly oriented their R&D investment and launch strategies toward the U.S. market. American patients benefitted through earlier access to new therapies, a broader range of treatment options, and sustained improvements in quality of life.
Today, America’s biopharmaceutical leadership and the health and economic benefits it brings are at risk. U.S. policymakers are considering or are already pursuing steps that would erode the very foundations of the country’s innovation ecosystem, including reductions in federal research funding, expanded price controls, policies that weaken intellectual property (IP) protection, and tariffs that would disrupt pharmaceutical supply chains. For patients, these policies would translate into reduced clinical trial access in the United States, slower development of new therapies, reduced treatment options—notably for rare and complex diseases—and delayed access to innovative medicines available elsewhere. As the international landscape shifts and other nations mobilize to strengthen their own biopharmaceutical capabilities, the lessons learned from Europe’s experience—wherein policy choices ultimately reduced drug R&D and innovation as well as timely patient access to new treatments—are increasingly important.
Just as the United States rose to global life sciences leadership as Europe’s position waned, China is likewise working feverishly now to supplant the United States.
America’s position as the global life sciences innovation leader, and the health and economic benefits that result from it, is not assured. It depends on sustaining policy choices that support innovation, investment, and patient access. Understanding the consequences of Europe’s past decisions is essential to ensuring that the United States avoids repeating its mistakes. That matters, because just as the United States rose to global life sciences leadership as Europe’s position waned, China is likewise working feverishly now to supplant the United States.
Europe’s Biopharmaceuticals Decline: A Cautionary Tale
Europe’s trajectory—from global biopharmaceutical leader to follower—offers a clear warning. Policies introduced beginning in the 1980s—including strict drug price controls, aggressive price negotiation tactics, and regulations that limited biotechnology research and entrepreneurship—steadily eroded the continent’s competitiveness. These choices created the opening through which the United States would emerge as the new global leader.
For decades, Europe dominated the biopharmaceutical landscape. Between 1960 and 1965, European firms produced 65 percent of all new medicines in the world. Even into the 1970s, European companies were introducing more than twice as many new drugs as did their U.S. counterparts (149 compared with 66).[1] And throughout the 1980s, fewer than 1 in 10 new medicines were launched first in the United States.[2]
As late as 1990, Europe remained the center of global pharmaceutical R&D activity: industry investment there was roughly 50 percent higher than in the United States.[3] At the beginning of the decade, Europe still accounted for half of global biopharma R&D investment.[4] And companies on both sides of the Atlantic held comparable shares (roughly one-third) of the global drug market. But the 1990s marked a turning point. Over that decade, Europe’s position steadily weakened. By 2004, its share of global pharmaceutical innovation had fallen to 18 percent, while the U.S. share had climbed to 62 percent.[5] Between 1990 and 2017, growth in U.S. pharmaceutical R&D investment was nearly twice that of Europe’s.[6] American biopharmaceutical firms, whose R&D spending in the 1970s amounted to only about half that of their European peers, were out-investing them more than threefold by the mid- to late-1990s.[7]
As the European Federation of Pharmaceutical Industries and Associations (EFPIA) noted in 2020, almost half of all new global therapies produced between 2014 and 2018 originated in the United States, compared with only one-quarter in Europe—a significant reversal from just 25 years prior.[8] By 2014, almost 60 percent of new drugs launched globally were introduced first in the United States—a pattern that reflected both the shift in where innovation was occurring and the fact that global companies increasingly chose the U.S. market for initial launch in order to recoup their R&D investments. For U.S. patients, this translated into earlier access to breakthrough medicines, greater availability of clinical trials, and more treatment options—including in areas of unmet medical need such as cancer, rare diseases, and complex chronic conditions.
Europe’s complex web of government-enforced pricing and reimbursement rules kept prices so low that companies sometimes questioned whether launching new therapies in certain European countries was economically viable.
Europe’s decline from the “world’s medicine cabinet,” and the corresponding rise of the United States, can be traced in large part to public policy choices. European governments adopted pricing and regulatory frameworks that constrained innovation, while the United States pursued more supportive policies. Analysts observed that Europe’s complex web of government-enforced pricing and reimbursement rules kept prices so low that companies sometimes questioned whether launching new therapies in certain European countries was economically viable.[9]
Price Controls
Beginning in the 1980s, many European nations implemented a broad array of pharmaceutical price control tools, including international (and regional) reference pricing systems, cost-focused health technology assessments, global prescribing budgets that held provider organizations accountable for exceeding spending budgets, profit controls that capped what insurers could pay for groups of similar drugs, and rules limiting the use of higher-priced medicines to hospitals.[10]
The following provides descriptions of how price controls were implemented in several European countries. In Germany, the Health Reform Act of 1989 introduced a national reference price system for reimbursable drugs to reduce rising pharmaceutical expenditures. The reform grouped drugs into three classes and established reimbursement ceilings for each class. Under this system, a drug’s reference price was computed as the average price of that drug and therapeutically similar products.[11] A U.S. Government Accountability Office (GAO) report explains that, in 1993, Germany implemented across-the-board price cuts on drugs not covered by the reference price system, as well as a reduction in nonprescription drug prices.[12] A study that examined the average prices per Defined Daily Dose (DDD)—the average daily dose for a drug—across eight therapeutic classes (beta-blockers, calcium antagonists, non-opiate analgesics, oral hypoglycemics, nonsteroidal anti-inflammatory drugs (NSAIDs), expectorants, coronary dilators, and systemic antibiotics) found that, as expected, each therapeutic class’s average price per DDD decreased immediately after the introduction of reference prices.[13]
In the Netherlands, the introduction of the 1996 Medicine Prices Act set maximum prices for prescription medicines based on the average price of similar drugs across a four-country reference basket. From 1996 to 2020, these were Belgium, France, Germany, and the United Kingdom.[14] In October 2020, the Netherlands implemented a revised international reference pricing rule under which Norway replaced Germany as a reference country in order to further reduce medicine prices, reflecting Norway’s lower drug price levels.[15]
For France, the GAO report characterizes post-1980 pharmaceutical regulation as relying on product-by-product drug price controls. GAO explained that drug prices were determined through a three-step process, which included drug safety and efficacy review and approval, a determination of the extent of a therapeutic advance of a drug compared to existing drugs, and a review of whether the drug was expected to offer benefits to the national economy. The government also limited price changes through blanket pricing directives, including one issued in 1991 that mandated a price decrease of 2.5 percent on all drugs.[16] Since 2013, pricing decisions have also incorporated a cost-effectiveness evaluation conducted by the Economic and Public Health Committee. Price setting also accounts for the price of comparator drugs, and price and coverage decisions are reviewed at least every five years.[17]
For Sweden, GAO reported that, in 1993, the government established a reimbursement system that determined drug reimbursement levels through reference pricing, either by setting the reimbursement price for brand-name drugs where equivalent generics exist or by directly setting the reimbursement price for drugs not under the reference pricing system. The reimbursable amount was set at the price of the lowest generic equivalent plus 10 percent.[18]
Variants of these mechanisms remain embedded in today’s systems, including fixed reimbursement prices in France and profit limits in the United Kingdom.[19] These policies have often been justified as a way to prevent drug pricing from outpacing inflation, even though economic efficiency would justify real price increases as pharmaceutical demand rises more than proportionately with income.[20]
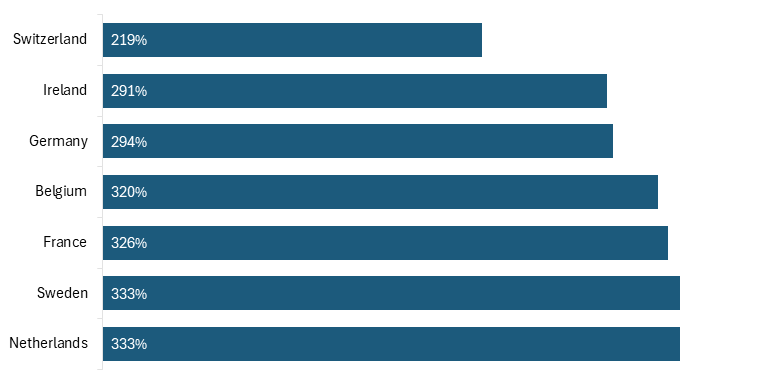
Taken together, these mechanisms have contributed to systematically lower pharmaceutical prices in many European markets relative to the United States, prompting claims that Europe has been “free riding” on U.S. biopharmaceutical innovation. To illustrate these price differences, a 2024 RAND report calculates each non-U.S. country’s drug price index relative to the United States, weighted by U.S. drug market shares. Using RAND’s price index data, the Information Technology and Innovation Foundation (ITIF) calculated U.S. prescription drug prices as a percentage of prices in selected European countries, illustrating the magnitude of price differences.[21] Following RAND’s methodology, ITIF reports U.S. drug prices relative to those in a comparison country fixed at 100 percent. On this basis, U.S. prices were 320 percent higher than prices in Belgium, 326 percent higher than France, 294 percent higher than Germany, 291 percent higher than in Ireland, 333 percent higher than the Netherlands and Sweden, and 219 percent higher than in Switzerland.[22] (See figure 1.)
Figure 1: U.S. prescription drug prices as a share of prices in selected European countries, 2022
 Notably, while U.S. brand name drug prices are higher than in other developed countries, the RAND report finds that U.S. unbranded generics—accounting for roughly 90 percent of prescription volume—were on average cheaper, at approximately 67 percent of prices in comparison countries, underscoring the extent to which price differences are concentrated in the innovative segment of the U.S. drug market.[23]
Notably, while U.S. brand name drug prices are higher than in other developed countries, the RAND report finds that U.S. unbranded generics—accounting for roughly 90 percent of prescription volume—were on average cheaper, at approximately 67 percent of prices in comparison countries, underscoring the extent to which price differences are concentrated in the innovative segment of the U.S. drug market.[23]
Consequences for European Patients
Europe’s drug price controls have come with substantial consequences for patient health. A 2004 analysis found that European patients faced delayed access to new therapies and that health outcomes suffered.[24] A study by the Galen Institute finds that out of 290 new drugs that became available between 2011 and 2018, 89 percent were available in the United States, compared with 62 percent in Germany, 48 percent in France and Switzerland, and 40 percent in Ireland. Zooming in on oncology, where timing is often critical, the gap was also significant: 96 percent availability in the United States, compared with 73 percent in Germany, 66 percent in France, 62 percent in Switzerland, and 51 percent in Ireland.[25] Further, a recent IQVIA study finds that among 329 drugs approved by both the U.S. Food and Drug Administration (FDA) and the European Union’s (EU) European Medicines Agency (EMA) between 2014 and 2022, 79 percent (260) were approved in the United States first, with U.S. approval occurring an average of 0.4 years earlier than in the EU.
A 2004 analysis found that European patients faced delayed access to new therapies, health outcomes suffered, research investment fell, and high-value pharmaceutical jobs disappeared.
More importantly for patients, time to availability—measured from regulatory approval to payer coverage—was longer in 25 of the 27 EU countries than in the United States, with 22 of 27 EU countries experiencing median delays of one year or longer for novel treatments.[26] For patients with serious or life-threatening illnesses, such delays are not abstract—they can mean prolonged reliance on older, less-effective treatments, fewer clinical options when disease progresses, and, in some cases, the loss of a meaningful chance for survival or improved quality of life.
As Lakdawalla and colleagues have shown, reductions in pharmaceutical spending today diminish the future stock of novel drugs, ultimately harming patients.[27] Their analyses projects that lowering U.S. drug prices toward European levels would reduce life expectancy for adults ages 55–59 by roughly two-tenths of a year.[28] Applied across the current U.S. population ages 55–59—approximately 21.3 million individuals—a reduction of two-tenths of a year in life expectancy per person would amount to more than four million life-years lost.[29] Thus, the evidence is clear: drug price controls harm patients worldwide.
Consequences for European Drug R&D and Innovation
Research shows that the long-term societal costs of those lost or delayed drugs outweigh the short-term benefits of reduced drug prices. A study published in Pharmacoeconomics shows that EU price controls have reduced European pharmaceutical firms’ R&D investments compared with American companies, resulting in 46 fewer new drugs and 1,680 fewer research jobs in the EU between 1986 and 2004.[30]
According to data provided by EFPIA, from 1999 to 2023, U.S.-headquartered firms produced 1.5 times as many new chemical entities (NCEs) or new biological entities (NBEs) as did their European counterparts, a gap that has continued to widen in recent years with U.S. companies producing more than 1.8 times as many drugs as EU firms did between 2014 and 2023. (See figure 2.)[31]
Figure 2: Number of new chemical or biological entities, 1999–2023[32]
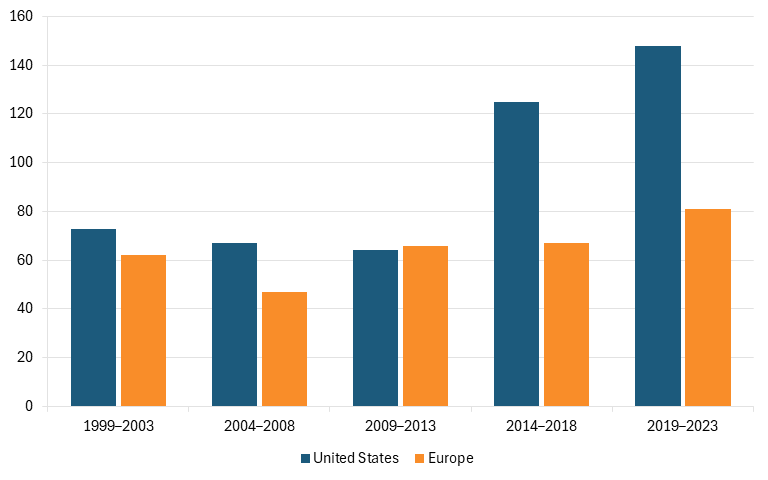
These findings align with a broader body of work on the impact of price controls on innovation. A study by Golec and Vernon finds that European price controls sharply reduced European firms’ incentives to invest in R&D.[33] In 1986, European companies invested 24 percent more in R&D than did U.S. firms. By 2004, this had reversed, with U.S. companies investing 15 percent more than their European counterparts. The analysis estimates that price controls reduced European R&D by roughly 20 percent between 1986 and 2004, resulting in 46 fewer new medicines and the loss of 32,000 R&D jobs.[34] A complementary analysis reaches similar conclusions. It estimates that, absent price controls, drug prices in Organization for Economic Cooperation and Development (OECD) countries would have been 35 to 45 percent higher, generating an additional $17 billion–$22 billion in annual R&D investment and yielding 10–13 more new medicines each year.[35]
More recent scholarship reinforces these findings. A 2024 study by Ho and Pakes argues that pharmaceuticals generate global benefits—the moment a drug is developed, patients worldwide can benefit from it—yet no international framework exists to align pharmaceutical pricing or public research contributions across countries. As a result, the study shows that the United States effectively subsidizes global drug development, in large part due to its higher prices.[36] The authors’ analysis of a hypothetical unified pricing model across high-income (OECD) countries shows that U.S. prices could fall by half, but only if prices in other nations rose substantially—for example, to 148 percent of current levels in Germany, 197 percent in France, 263 percent in Italy, and 287 percent in Spain.[37]
Price controls do not only affect present-day production and availability of new medicines; they also shape the therapies available to future generations. By dampening returns on innovation, these policies limit the development of new treatments for diseases such as cancer, Alzheimer’s, heart disease, and diabetes. Many countries benefit from medical breakthroughs generated elsewhere, but that reliance ultimately comes with costs of its own.
Other Factors Behind Europe’s Decline
Price controls were not the only contributing factor behind Europe’s decline. Regulatory philosophy also mattered. The United States embraced “innovation-principle”-focused regulation, seeking to avoid unnecessary regulatory burdens and adopting policies supportive of technological progress. Europe, by contrast, tended to follow a “precautionary principle,” which places the burden on innovators to demonstrate the absence of potential harm before new technologies can be widely adopted, even when scientific evidence does not permit a complete evaluation of risk, imposing further constraints on an already heavily regulated industry.[38]
Other dimensions of Europe’s policy environment further weakened competitiveness. A notably cautious approach to mergers limited firms’ ability to achieve the scale increasingly required for effective drug development. Even relatively modest consolidations—such as the 2006 Schering-Bayer merger, which created only the world’s 12th-largest drug company when Bayer acquired the Berlin-based pharmaceutical company Schering—met regulatory skepticism.[39] This stance stands in contrast to the historical reality that Europe originally produced industry champions precisely because earlier companies were able to grow large enough to compete globally.[40] More broadly, Europe’s policy environment has historically been less conducive to high-risk entrepreneurship and rapid firm scale-up, limiting the emergence of globally competitive biopharmaceutical companies.[41]
Regulatory culture also played a role. As Arthur Daemmrich has argued, the way countries balance patient protection with consumer choice and empowerment has profound implications for the competitiveness of their domestic industries.[42] Differences in regulatory philosophy—such as responses to new diseases, rules around compassionate use, integration of biomarkers, and other aspects of consumer-oriented drug development—help explain diverging national trajectories. He suggested that “the predictability of centralized regulation based on a tight regime of quantified clinical trials in the United States coupled to the emergence of a focus on consumers and their access to drugs ultimately benefited firms operating in that country over their German counterparts.”[43]
Taken together, this overview of Europe’s lost pharmaceutical leadership offers a clear warning: U.S. policymakers should be extremely cautious about adopting the very policies that contributed to Europe’s decline.
U.S. Biopharmaceutical Leadership and Its Foundations
Today, the United States is the global leader in biopharmaceuticals, with new drug development substantially greater than that of the rest of the world. In addition to a large domestic market, which accounts for 42 percent of global drug sales, several factors have contributed to U.S. leadership in biopharmaceutical innovation—and this leadership has translated into American patients consistently being among the first worldwide to have access to breakthrough therapies.
Limited Drug Price Regulations
There exists an inherent linkage between biopharmaceutical companies’ ability to earn profits and their ability to invest in R&D to fund the next generation of innovation, and most academic studies find strong positive links between industry returns and R&D investments.[44] One study, for example, finds a statistically significant positive relationship between a biopharma firm’s profits from the previous year and its R&D expenditures in the current year.[45] Another shows that sales revenue from previous periods has a significant, positive impact on current-period biopharma R&D.[46] Other work has found that the pharmaceutical firms with the greatest sales have the largest R&D investments.[47] And this can translate into launched drugs, as a 2015 paper finds that every $2.5 billion of additional biopharmaceutical revenue leads to one new drug approval.[48]
Had the United States adopted similar price controls to Europe, estimates suggest a loss of 117 new drugs and 4,368 research jobs.
If the United States were to impose drug price controls similar in magnitude to those adopted in Europe, pharmaceutical companies would likely reduce R&D investments, resulting in fewer new treatments and cures. For instance, estimates suggest that, had the United States adopted similar price controls to Europe, it would have lost approximately 117 new drugs and 4,368 research jobs.[49] A Health Affairs study further estimates that such a policy would hurt future U.S. generations and impose a cost of $8 trillion on the United States.[50] These potential losses underscore how central innovation-friendly policies have been to U.S. biopharmaceutical leadership. Several additional factors have also contributed to that leadership, including world-class research institutions, innovative regulatory frameworks, and strong IP protections.
Strong Research Institutions
A major contributor to U.S. leadership has been the strength of its research institutions. The National Institutes of Health (NIH), with a budget of $48 billion in 2025, up from $11 billion in 1996, supports roughly 300,000 scientists at more than 2,500 research institutions nationwide.[51] In parallel, the Defense Advanced Research Projects Agency (DARPA) has played a catalytic role by funding high-risk, high-reward biomedical research, including in areas such as mRNA vaccine platforms, rapid diagnostics, advanced biologics manufacturing, and pandemic preparedness.[52] Complementing the NIH’s foundational science and modeled on DARPA’s strategy, the Advanced Research Projects Agency for Health (ARPA-H), established in 2022 with a current budget of $1.5 billion, aims to accelerate high-impact solutions to some of the toughest health challenges.[53] Complementing federal research support, the United States benefits from a robust network of academic research institutions and technology-transfer policies—notably the 1980 Bayh-Dole Act—that have facilitated the translation of basic science into commercial ventures and strengthened industry partnerships.[54]
Innovative Pharmaceutical Regulation Legislation
Policies favorable to the U.S. biopharmaceutical industry include the Bayh-Dole Act, the Orphan Drug Act (enacted in 1983), and the Prescription Drug User Fee Act (PDUFA) of 1992 (and its subsequent reauthorizations).
The Patent and Trademark Amendments Act of 1980 (also known as the Bayh-Dole Act) transformed the way federally funded research moves from lab to market by creating a framework for public-private collaboration. The act allowed universities to patent and license inventions arising from federal grants—rather than leaving those rights with the government—creating strong incentives for academic institutions to commercialize discoveries.[55] The results have been truly dramatic: university patents increased from 380 in 1980 to 3,088 by 2009, and university licensing income increased from $0.6 billion to $2.3 billion between 1995 and 2015.[56] Moreover, the number of university-based start-ups nearly doubled, from 400 in 2001 to 760 in 2013.[57] Bayh-Dole is widely viewed as one of the most successful technology-transfer laws in history, playing a key role in fostering the growth of vibrant regional tech ecosystems across the United States.[58]
The Orphan Drug Act of 1983 sought to incentivize the development of drugs for rare diseases, which may not be economically viable without special incentives, through benefits such as tax credits, grants, and extended market exclusivity to pharmaceutical companies that develop and bring to market drugs for these rare diseases.[59] From 1983 to 2022, there were 1,122 approvals of orphan-designated products, including new molecules, indication, and formulation approvals.[60]
Congress passed PDUFA in 1992, specifying performance goals for the FDA in the form of quicker drug application review times in exchange for authorizing the FDA to collect fees from drug manufacturers to enhance resources that would expedite the review process. A 2005 study found that PDUFA substantially accelerated the FDA’s drug review process. Instead of a counterfactual 16 percent reduction in approval times in the absence of PDUFA from 24.2 to 20.4 months, approval times decreased 42 percent, from 24.2 to 14.2 months, between 1991 and 2002. Therefore, PDUFA accounted for approximately 62 percent of the decline in drug approval times during this period.[61]
The FDA’s Breakthrough Therapy Designation (BTD) program, launched in 2012, provides another example of supportive legislation. Designed to expedite the development of drugs targeting serious conditions with unmet medical needs, the program has been instrumental in shortening clinical development timelines and enabling faster market entry for life-saving therapies, while maintaining rigorous safety standards. The BTD program’s success offers valuable lessons for policymakers seeking to balance timely access and safety in approving lifesaving therapies.[62]
In addition, as noted, the U.S. market has historically limited government drug price regulation, which has played an important role in spurring domestic pharmaceutical innovation and greater availability of medicines for patients in the United States.[63]
Robust IP Protection Regime
The drug development and approval process is expensive, lengthy, and risky. While estimates vary widely across therapeutic areas, recent figures suggest that the R&D cost of bringing a new drug to market could be up to $2.83 billion (uncapitalized), factoring in pre- and post-approval R&D such as new indications, patient populations, and dosage forms—or, capitalized at an annual rate of 10.5 percent, up to $4.04 billion.[64] Drug development also takes 10–15 years, and few drugs ever reach the market. Namely, fewer than 1 in 10 drugs that enter Phase I clinical trials eventually make it to market.[65] A pharmaceutical company developing a new drug incurs the full costs of R&D, but it often does not capture the full benefits because as soon as a new drug is released, other firms may quickly replicate it. (An example of this would be how competition rapidly drove down the cost of hepatitis-C treatments.)[66] These knowledge spillovers highlight the need for incentives that encourage pharmaceutical companies to invest in the necessary R&D to create a drug in the first place. To encourage socially desirable levels of R&D, a commonly used policy tool is patent protection.[67]
Patents, a form of IP protection, grant exclusive rights to commercialization of an invention for a specific period, typically 20 years. In the pharmaceutical industry, where drug development is lengthy and costly, patents are especially important to stimulate R&D investments. Patents provide time-limited market exclusivity that enable pharmaceutical companies to recoup investment costs, helping offset the high risks and costs of R&D and support continued investment.[68]
From 2014 to 2013, U.S. drug companies produced more than 1.8 times as many new drugs as EU firms did.
Using data from the Carnegie Mellon Survey on Industrial R&D, a 2008 study provides evidence that patent protection leads to higher private returns on R&D, which in turn stimulates future R&D investments in the pharmaceutical industry.[69] Another study of 60 countries from 2000 to 2013 finds that patents are associated with the earlier release of new drugs.[70] And a third study using data on the launches of 642 drugs in 76 countries from 1983 to 2002 finds that a higher length and strength of patent protection significantly accelerates drug diffusion, spurring market entry in high-income countries.[71] It also appears that IP protection on research tools in biomedical innovation seldom hinders the pursuit of scientifically promising projects, meaning patents do not impede future innovation.[72]
Current U.S. Policy Missteps
While strong IP protections and a general absence of government-imposed price controls have long supported U.S. biopharmaceutical innovation, recent proposals to dictate drug prices or weaken IP protections threaten this leadership. These efforts are typically motivated by the desire to lower drug prices. Yet, they reflect a misunderstanding of the trade-offs involved and risk undermining the very ecosystem that produces novel, frequently life-saving therapies.
Drug Price Controls
U.S. policymakers face growing pressure to make health care more affordable. At the heart of this debate, when it comes to drug prices, is a well-known trade-off: lower prices today versus fewer new drugs tomorrow.[73] Both Democratic and Republican administrations have pursued forms of price setting—most notably the Biden administration’s Inflation Reduction Act (IRA) and the Trump administration’s proposed Most-Favored Nation (MFN) policies. Despite their differences, these approaches share an underlying issue: they attempt to reduce spending by suppressing returns on innovative medicines, a strategy that other countries have adopted at the cost of weaker biopharmaceutical ecosystems.
Although medicines account for just 14 percent of U.S. health care spending, proponents of price controls argue that consumers need protection from rising prices.[74] But this overlooks the fact that drug prices—after rebates and discounts—have been falling for years, with much of the savings captured by intermediaries such as pharmacy benefit managers rather than patients, as drug developers’ share of each dollar spent on drugs fell from 66.8 percent in 2013 to 49.5 percent in 2020.[75] Meaningful reforms must therefore address the entire pharmaceutical pricing supply chain—from manufacturers to insurers, pharmacy benefit managers, and pharmacies—rather than targeting drug innovators and manufacturers alone.
Moreover, while it is true that Americans are spending more on health care, they are not necessarily spending more on drugs. For instance, according to U.S. Bureau of Labor Statistics data, Americans’ reported expenditures on health insurance (in nominal dollars) increased by over 127 percent from 2006 to 2024, while their total health care expenditures increased 98 percent. While consumers’ expenditures on drugs did increase 35 percent during that time, that was barely more than one-third of the overall increase in health care expenditures. Moreover, in real dollars, consumers’ expenditures on drugs actually fell by 7 percent over that timeframe. (See figure 3.)[76]
Figure 3: Percentage change in U.S. consumers’ health care expenditures, 2006–2024 (real dollars)[77]
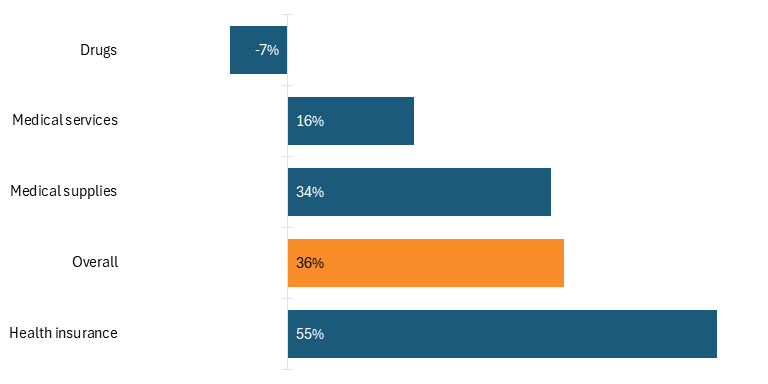
The Inflation Reduction Act
In August 2022, Congress passed the IRA, which introduced, for the first time, federal authority for the Centers for Medicare & Medicaid Services (CMS) to control prices for selected drugs targeting a wide range of diseases, including diabetes, heart disease, and cancer.[78] While the intent was to make medicines more affordable for seniors, the early effects of the policy suggest significant risks for innovation. Since the IRA’s drug-pricing framework was first drafted in 2021, venture funding for small-molecule R&D has fallen nearly 70 percent.[79] Moreover, a PhRMA survey of biopharmaceutical companies finds that 78 percent of them expect to cancel early-stage small-molecule projects.[80] Nearly two-thirds reported that they plan to shift R&D away from small molecules entirely. This matters because small-molecule drugs are essential for conditions including neurogenerative and cardiovascular diseases that disproportionately affect older Americans. Another study finds the number of therapies in Phase I and Phase II of development declined 35 percent from 2021 to 2023 among small and midsize biotech companies, with analysts projecting fewer FDA approvals five to six years from now.[81]
Since the IRA’s drug-pricing framework was first drafted in 2021, venture funding for small-molecule R&D has fallen nearly 70 percent.
Estimates of long-run effects are even more concerning. A 2021 University of Chicago study projects that IRA-style price controls would reduce pharmaceutical R&D by 45 percent between 2021 and 2039, leading to 254 fewer new medicines and large long-term mortality impacts.[82] A 2023 U.S. Chamber of Commerce report further shows that price controls could sharply reduce funding for clinical trials. The United States outperforms OECD peers by 42 to 220 percent in clinical research activity. IRA-style price controls could cut early-phase projects by 50 percent, including deep reductions in research on biologics (59 percent) and cancer (54 percent).[83] In short, the IRA’s approach risks weakening the R&D engine that has enabled the United States to lead the world in life sciences innovation.
Most-Favored Nation Pricing
In May 2025, the Trump administration issued an executive order directing the Department of Health and Human Services (HHS) to pursue an MFN pricing approach.[84] The objective was to enable Americans to buy medicines at the lower prices charged in peer countries.
But MFN would amount to importing other countries’ price controls into the United States. Unlike Medicare price-setting under the IRA, which applies to selected lists of drugs, MFN would effectively cover all medicines. ITIF has explained how CMS’s plans to implement MFN approaches through its proposed Global Benchmark for Efficient Drug Pricing (GLOBE) and Guarding U.S. Medicare Against Rising Drug Costs (GUARD) models are misguided.[85]
Moreover, these proposals arrive at a time when the United States faces rising competition from China, which has adopted an aggressive national strategy to expand its biopharmaceutical capabilities. Imposing broad price controls at home would weaken U.S. competitiveness just as a major rival accelerates its ambitions.[86] Rather than adopting the pricing practices of countries that invest less in biopharmaceutical innovation, U.S. policy should focus on ensuring that other high-income countries contribute more fairly to global pharmaceutical R&D, rather than relying on U.S. consumers to shoulder the costs. Further, U.S. policy should expand public-private partnerships (PPPs) in order to strengthen domestic manufacturing and advance new technologies that reduce production costs.
Pharmaceutical Tariffs
The Trump administration’s proposal to impose a 100 percent tariff on brand name drugs unless manufacturers build new production facilities in the United States is harmful to American patients. It would not meaningfully boost domestic production in the short term, but it would immediately make medicines more expensive for U.S. consumers, invite foreign retaliation, and weaken the innovation system that produces new therapies.
A 100 percent tariff would sharply increase the import cost of brand-name medicines, hurting American patients, as many drugs they take every day are imported or depend on imported ingredients. ITIF warned in its May 2025 comments regarding the Section 232 investigation that tariffs could “considerably increase the price Americans pay for medicines.”[87] Patients would pay more, and many would end up delaying or forgoing treatment.
This is because, while the United States has capabilities in pharmaceutical R&D and final drug production, it remains reliant on foreign sources for active pharmaceutical ingredients (APIs), the components in medications that are biologically active and directly enable drugs’ therapeutic effects. Reshoring the pharmaceutical supply chain (i.e., building domestic manufacturing capacity for APIs) represents a critical national security need, but this is a process that takes time, and reshoring the manufacturing supply chain cannot happen overnight. Foreign supply chains are essential to U.S. pharmaceutical access, particularly for APIs and generic drugs. In the United States, where 9 out of 10 prescriptions dispensed are for generic drugs, India supplies roughly 40 percent of those drugs.[88]
Tariffs would also harm biopharmaceutical innovation. R&D is risky and costly, and pharma companies rely on global markets to recoup those investments. By cutting off or distorting access to those markets, tariffs undermine the incentives to invest in the next generation of treatments. Further, pharmaceutical supply chains are global, and novel medicines rely on highly integrated international networks. These networks reflect comparative advantage and the need for specialized facilities—and they deliver safe and affordable medicines at scale. A tariff that seeks to force domestic production ignores this reality and would instead fracture supply chains, increase bottlenecks, and raise the risk of shortages.
Foreign supply chains are essential to U.S. pharmaceutical access, particularly for APIs and generic drugs.
There is also the threat of retaliation for U.S. tariffs. If the United States imposes blanket tariffs, other countries will hit back. U.S. drug exports would be at risk, and American firms could face steep countermeasures in their fastest-growing markets. A policy intended to strengthen U.S. manufacturing could end up shrinking U.S. global market share instead. If the goal is to reduce reliance on foreign drug production, there are better tools than tariffs. As explained subsequently, the United States should invest in advanced manufacturing, support domestic capacity with targeted subsidies and tax credits, and encourage other countries to pay their fair share for innovative drugs.
Proposed Cuts to NIH Funding
NIH critically underpins the strength of America’s biopharmaceutical innovation ecosystem. It sustains the nation’s research capacity, supports a skilled scientific workforce, and drives medical breakthroughs that power U.S. economic and strategic leadership and protect national security.
Unfortunately, the Trump administration’s proposed FY 2026 budget would slash NIH funding by approximately 40 percent, from about $48 billion in 2025 to roughly $27 billion in 2026, an unprecedented and misguided reduction that would also eliminate entire institutes, dismantling vital core infrastructure built over decades.[89]
Already, $1.7 billion in funds have been withheld, and over 2,200 grants totaling $3.8 billion have been canceled, prompting universities to freeze hiring, delay clinical trials, and scale back or shut down laboratories.[90] Modeling studies show that an ongoing 10 percent cut in NIH funding would reduce new drug launches by 4.5 percent annually—the equivalent of losing roughly two new lifesaving medicines each year.[91]
The implications are stark: cuts of this magnitude could hollow out America’s biopharmaceutical capacity, reduce its scientific workforce at a time when other countries are actively seeking to attract U.S.-based scientists, disrupt the innovation pipeline, and weaken national preparedness against health emergencies.
Modeling studies show that an ongoing 10 percent cut in NIH funding would reduce new drug launches by 4.5 percent annually—the equivalent of losing roughly two new lifesaving medicines each year.
Bayh-Dole March-In Rights
The Bayh-Dole Act of 1980 created a framework for public-private collaboration, allowing universities to patent and license inventions arising from federal grants and becoming one of the most successful technology-transfer laws in modern history.[92]
However, despite its success, in recent years, some policymakers have sought to reinterpret one provision of the act: march-in rights, which allow the government, under limited circumstances, to require patent holders to license their inventions to others.[93] The authors of the law made clear that this authority was intended to ensure that inventions were actually developed and commercialized—not to regulate prices. Senators Birch Bayh and Bob Dole explicitly rejected the idea that march-in rights should be used to ensure “reasonable prices,” and the statute contains no such language.[94]
Despite this, proposals have emerged to use march-in rights as a drug-price control tool. Senator Angus King (I-ME), for example, has proposed legislation that would require compulsory licensing “whenever the price of a drug, vaccine, or other medical technology is higher in the U.S. than the median price charged in the seven largest economies that have a per capita income at least half the per capita income of the U.S.”[95] The Biden administration also floated draft guidance in 2023 that would have permitted governmental use of march-in rights on the basis of the resulting price of a product, though it ultimately did not finalize the policy.[96] Using march-in rights as a price-control mechanism would undermine partnerships among universities, federal laboratories, and private companies that have supported biopharmaceutical innovation.
The first Trump administration directed the National Institute of Standards and Technology (NIST) to review federal policies aimed at bolstering the return on federal R&D investments. That review reaffirmed that march-in rights are a last resort and have never been used since the act’s passage to control prices.[97] NIH similarly determined that using march-in rights to control drug prices “was not within the scope and intent of the authority.”[98] Fortunately, the second Trump administration has signaled no plans to use march-in rights for this purpose. In January 2025, HHS Secretary Robert F. Kennedy Jr. stated that employing march-in rights to lower drug prices “would not be an appropriate use of march-in rights.”[99]
Constructive Policy Alternatives
Certainly, drug affordability matters. But there are more effective ways to address it while stimulating innovation than through price controls such as MFN or limits on IP protections.
A more constructive strategy for strengthening U.S. biopharmaceutical R&D and manufacturing—while addressing systemic cost pressures—would be to expand PPPs that accelerate technological innovation. This mirrors the conclusion of the April 2025 National Security Commission on Emerging Biotechnology report, which recommends that the federal government invest at least $15 billion over five years to catalyze private capital in U.S. biotechnology.[100]
A new generation of therapies, including cell and gene therapies, will also require novel manufacturing platforms and facilities. The United States should focus on leading development of these next-generation technologies. Doing so will also depend on supportive tax policy, regulatory and permitting policy, investment in education and workforce skills, and sustained R&D funding. These kinds of forward-looking policies—not price controls or tariffs—are what will help revitalize U.S. drug manufacturing, reverse pharmaceutical trade deficits, and ensure that patients have access to innovative and affordable drugs.
A new generation of therapies, including cell and gene therapies, will also require novel manufacturing platforms and facilities. The United States should focus on leading development of these next-generation technologies.
As the Trump administration considers tariff policy and broader trade negotiations, pharmaceutical cost sharing must also be part of the agenda. If other high-income countries want access to cutting-edge American medicines, they should contribute a fairer share to their development rather than rely on rigid price controls that shift the burden onto U.S innovators and patients. The only way to realign incentives is for the United States to credibly signal that it expects more equitable participation—a logic similar to calls for shared financing of other global public goods, such as collective security. MFN-style drug pricing would undermine this objective by importing foreign government price controls into the U.S. market. Rather than encouraging other countries to contribute more, it would entrench globally suppressed prices, weakening incentives for innovation and eroding the system on which patients worldwide depend. The better path is to make drug pricing a core issue in ongoing trade negotiations and ensure that continued progress in medicine is supported by shared international investment. Policymakers can protect patients, taxpayers, and national security alike by pursing policies that reward science and save lives.
Europe’s Efforts to Reclaim Biopharmaceutical Leadership
Once leadership in an advanced technology industry such as biopharmaceuticals is lost, it is extremely difficult to rebuild. Europe is now attempting to regain its competitiveness through a series of efforts, taking advantage of U.S. policy missteps that have created a window of opportunity.
EU-Level Policies
The European Union has placed biotechnology at the center of its renewed competitiveness strategy. The Competitiveness Compass, launched in January 2025, aims to “restore Europe’s dynamism and boost our economic growth” by closing the innovation gap, decarbonizing the economy, and reducing dependencies.[101] It fosters a more friendly environment for start-ups, facilitates the adoption of new technologies by large companies, streamlines cross-border business activities, and accelerates technological development.
The proposed EU Biotech Act strengthens this framework by optimizing the research ecosystem, funding multi-country clinical trials under a One Health approach, and creating faster market-access pathways.[102] Complementing this, the Choose Europe to Start and Scale campaign (launched in May 2025) targets the “double valley of death” faced by high-growth firms, making Europe a more attractive base for biotech scale-ups and unicorns.[103] The EU is also taking steps to deepen research excellence through the “Choose Europe” initiative, an effort to attract global research talent.[104]
Once leadership in an advanced technology industry such as biopharmaceuticals is lost, it is extremely difficult to rebuild.
The EU’s financial backing reflects the sector’s strategic importance: the life sciences industry already contributes €1.5 trillion to the EU economy and supports 29 million jobs.[105] According to the European Innovation Council (EIC) Impact Report of 2023, more than €350 million has been invested in biotechnology and biomanufacturing through the Horizon Europe EIC portfolio.[106] Under the Competitiveness Compass, an additional €300 million has been mobilized to stimulate procurement of life sciences innovations addressing areas including next-generation vaccines and affordable cancer solutions.[107]
Collaboration between public institutions and industry remains central to the EU’s biotechnology vision. The Bio-Based Industries Joint Undertaking (BBI-JU), a major EU PPP, co-funds research and innovation projects that advance bio-based industrial transformation.[108] These partnerships align industry capital with public investment to accelerate commercialization, de-risk R&D, and promote Europe’s leadership in sustainable biomanufacturing and circular-economy technologies.
Through the Competitiveness Compass and related measures, the EU is simplifying regulatory processes and promoting consistent tax treatment for R&D activities. Although fiscal tools vary across member states, the shared objective is to reduce administrative friction, improve cross-border capital mobility, and lower barriers to firm entry and growth within the single market—aiming to create a more integrated environment for emerging life sciences companies.
The forthcoming EU Biotech Act underscores that “harmonized and internationally competitive IP protection rules for biotechnology and biomanufacturing patents are essential for the development of the industry.”[109] It calls for a common clinical-trials framework that incorporates real-world evidence and tasks the European Commission with assessing IP performance and recommending reforms. Framed as efforts to improve predictability and interoperability, these initiatives are intended to modernize patent and data-use regimes and strengthen the EU’s global competitiveness in biotechnology.
Moreover, the EMA is undergoing significant reforms, seeking to improve regulatory efficiency. Some of the changes include simplifying its scientific committee structure to both facilitate faster drug review and free up scientific resources to strengthen support for drug developers, creating frameworks for non-standard drug categories, and speeding up approval timelines for post-approval drug changes.[110]
In addition to EU-level efforts to strengthen biopharmaceutical competitiveness, several member states have launched national strategies that pursue the same objective. Some of these initiatives are outlined ahead and summarized in table 1.
Table 1: Biopharmaceutical innovation support policies employed by European countries
|
Policy |
France |
Germany |
Ireland |
The Netherlands |
Sweden |
United Kingdom |
|
National Life Sciences/Biotech Strategy |
✓ |
✓ |
✓ |
✓ |
✓ |
|
|
Talent Attraction Program |
✓ |
✓ |
✓ |
✓ |
* |
✓ |
|
R&D Tax Incentives |
✓ |
✓ |
✓ |
✓ |
✓ |
✓ |
|
IP Box Regime |
✓ |
✓ |
✓ |
✓ |
||
|
Institutional IP Ownership of Academic Inventions |
✓ |
✓ |
✓ |
✓ |
||
|
“Fast-Track” Scheme for Mononational Clinical Trials |
✓ |
✓ |
✓ |
|||
|
Health Data Systematically Available for R&D |
✓ |
✓ |
✓ |
✓ |
✓ |
|
|
Utility Model/Short-Term Patent Regime |
✓ |
✓ |
✓ |
|||
|
Government-Supported Life Sciences Clusters |
✓ |
✓ |
✓ |
✓ |
✓ |
* While Sweden does not have a branded talent attraction program, in 2025, the nation allocated 25 million SEK (approximately $2.8 million) to attract foreign researchers.
Germany
Germany anchors biotech within its cross-government Future Research & Innovation (R&I) Strategy (Zukunftsstrategie Forschung und Innovation), which orients federal R&I policy toward technological sovereignty, faster translation, and stronger start-up pathways.[111] The strategy functions as a “compass” to align ministries and instruments, including targeted life sciences measures. This pro-innovation orientation has also been reflected on the market-access side, with Germany moving away from international reference pricing—through the Medical Research Act of 2024—in an effort to strengthen the country’s attractiveness for biopharmaceutical R&D and manufacturing.[112] On the sectoral side, the National Bioeconomy Strategy—implemented primarily by the Federal Ministry of Education and Research (BMBF)—explicitly funds bio-based and biotech R&D.[113] To move ideas into firms, BMBF’s GO-Bio family (GO-Bio, GO-Bio initial/next) supports very early and translational life science projects through structured, two-phase support focused on commercialization readiness.[114]
Dr.Jasmina Kirchhoff, project manager for Pharmaceutical Location Germany at the Institut der Deutschen Wirstchaft, an economic research institute in Germany, believes that for Germany to improve its position in global biopharmaceutical innovation over the next decade, two policy areas need to be addressed: improving both translation and location factors from an industrial policy perspective. Regarding the first policy area, Dr. Kirchhoff has explained that, even though the breadth of local knowledge is strong, with clusters of excellence at universities and institutes that are among the best in the world, translation remains a challenge for Germany. For this excellence to be better reflected in marketable innovations, she has argued that several measures are needed: (i) modernizing the confusing regulatory framework that hinders spin-offs; (iii) strengthening the venture capital (VC) market; (iii) increasing digitization through improved infrastructure, including the rapid development of the European Data Health Space (EHDS), and the simplification of data protection; and (iv) reliable regulation that rewards innovation. Regarding the second policy area, she has noted that, from an industrial policy perspective, structural reforms are needed to promote private investment and innovation in growth sectors. This includes, among other factors, adapting regulations that inhibit innovation, further developing the European single market, improving corporate taxation, finding solutions to the shortage of skilled workers, and adjusting non-wage labor costs.
Germany has been moving away from international reference pricing—through the Medical Research Act of 2024—in an effort to strengthen the country’s attractiveness.
Germany’s overall R&D effort reached €121.4 billion in 2022 (R&D intensity representing 3.13 percent of gross domestic product (GDP)), with the federal strategy reaffirming a 3.5 percent target—resources that underpin mission-driven areas including health and biotech.[115] Within biotech-focused lines, BMBF’s GO-Bio initiative typically finances early life sciences projects (e.g., individual awards of €100,000–€1 million, depending on phase and call), while Clusters4Future and legacy Leading-Edge Cluster competitions have committed multi-year funds for regional health/biotech consortia (e.g., approximately €5 million per year for awarded clusters, with federal/state/industry co-investment).[116] BMBF’s Bioeconomy International line alone funded 142 projects in the amount of €81 million, illustrating dedicated biotech/bio-based investment beyond domestic calls.[117]
Germany institutionalizes PPPs through federal cluster programs and research centers that couple public labs and hospitals with companies. The Munich Biotech Cluster (m4)—supported under BMBF’s cluster initiatives—formalized more than 100 partners across academia and industry to advance targeted therapies, with federal backing to accelerate clinical translation.[118] Federally co-funded German Centers for Health Research (DZG) (e.g., DKFZ, DZD, DZHK, DZIF) run joint innovation funds and multicenter translational programs that pool university clinics and institutes with industry collaborators—initiatives that are explicitly designed to de-risk cross-disease, cross-site projects.[119]
Germany’s R&D tax incentive (Forschungszulage) provides a tax credit on eligible R&D costs for all tax-paying companies, complementing project grants.[120] The Federal Ministry of Finance notes the scheme’s role in strengthening the investment climate, with the law expanded in 2024 (Growth Opportunities Act) to broaden the assessment base and enhance SME support (e.g., through higher caps/percentages and recognition of additional cost categories).[121]
Biotech IP in Germany is protected through national patents (German Patent Act) and European/Unitary Patent routes; Germany ratified the Unified Patent Court system and has participated in it since its launch in June 2023, enabling centralized enforcement across participating EU states.[122] Germany also offers Utility Models (Gebrauchsmuster)—rapid, register-based protection (initial 3-year periods, extendable to 10)—which are useful for incremental biotech devices or tools.[123] For medicines and plant-protection products, supplementary protection certificates (SPCs) extend effective patent life under EU regulations, administered in Germany by the DPMA with published examination guidance.[124]
France
France’s “Innovation Santé 2030” (Health Innovation 2030) embeds biotech at the core of national competitiveness, with significant investments, dedicated acceleration strategies, and regulatory streamlining for clinical research and market access.[125] The plan prioritizes biotherapies and biomanufacturing, digital health, and preparedness for emerging infectious diseases, and it sets concrete goals for bioproduction, scale-up, and talent attraction. It also creates new instruments and governance (e.g., the Health Innovation Agency) to coordinate funding and simplify the innovation pathway. France 2030 further specifies an acceleration strategy for “biothérapies et bioproduction de thérapies innovantes,” aiming to make the country a European leader in developing and producing new biomedicines.[126]
The Health Innovation 2030 blueprint states that more than €7 billion is to be mobilized for health innovation, including €2 billion in public funding over five years for three strategic fields —biotherapies/bioproduction, digital health, and EID-CBRN—with the biotherapies pillar alone targeting €800 million in public plus €2 billion in private investment.[127] Additional France 2030 lines continue to finance life sciences infrastructures, bioclusters, university-hospital institutes, and preclinical/clinical capacity. Illustrative awards (e.g., genomics PEPRs) have also been announced by national research bodies under France 2030.
France is deploying bioclusters and university-hospital institutes (IHU) to concentrate academia–hospital–industry capacity and accelerate translation. Government roadmaps note the establishment of multiple new IHUs and bioclusters under the Health Innovation 2030 component of France 2030, explicitly to “bring together” innovation stakeholders on single sites and catalyze PPP-style collaboration from fundamental research through clinical development and industrialization.[128] France has also launched the “Choose France for Science” platform, an initiative aimed at enabling international researchers to continue their research in France amid weakening science environments abroad.[129]
The flagship Research Tax Credit (Crédit d’Impôt Recherche, CIR) is available to companies incurring eligible R&D expenditures. Official guidance highlights its scope and mechanics, and OECD documentation summarizes the headline rates (30 percent up to €100 million of eligible R&D, then 5 percent).[130] Widely used across sectors, including healthcare, the CIR has been maintained in recent finance bills and is frequently debated in the press (e.g., coverage of its scale around €7 billion–€8 billion annually). Start-ups may also access the “Jeune Entreprise Innovante” (JEI) regime, which provides exemptions on employer social security contributions for R&D staff and, for firms created before December 31, 2023, certain corporate-income-tax and local-tax reliefs. Rules have been updated since 2024 and are detailed on Unions de Recouvrement des Cotisations de Sécurité Sociale et d'Allocations Familiales (URSSAF) and Service-Public portals.[131]
France has launched the “Choose France for Science” platform aimed at enabling international researchers to continue their research in France amid weakening science environments abroad.
Companies can protect biotech inventions via French national patents (INPI), European patents, and, since June 2023, the Unitary Patent with centralized enforcement before the Unified Patent Court. French and EU pages explain how the Unitary Patent complements national rights.[132] For life sciences products, SPCs administered by INPI extend effective exclusivity following marketing authorization. INPI’s resources and the Ministry for the Economy’s guidance cover filing routes, searchable patent/SPC data, and strategic coexistence of national and unitary rights.
Ireland
Although Ireland does not yet have a comprehensive life sciences strategy, the Programme for Government 2025—a plan detailing the Irish government’s objectives over the next five years—outlines a clear commitment to developing a new National Life sciences Strategy.[133] The government has already solicited public input, including from industry stakeholders, to help ensure that the strategy is innovation friendly, fit for purpose, and aligned with the country’s competitiveness goals. This process is intended to support a more unified government approach and strengthen the long-term position of Ireland’s life sciences sector.
Ireland is the third largest pharmaceutical exporter in the world. Accounting for roughly 10 percent of global pharmaceutical exports, it is home to 19 of the world’s top 20 pharmaceutical companies.[134] The value of Ireland’s pharmaceutical exports is calculated at €80 billion a year.[135] Ireland is an attractive destination for global pharmaceutical companies due to its favorable tax policies, English-speaking workforce, and strong compliance record. Ireland’s corporate tax rate of 12.5 percent is relatively low compared with the global average of 23.5 percent.[136]
In 2023, Ireland’s gross expenditures on R&D reached €8.1 billion, with the government accounting for roughly €1 billion and the remainder originating from the EU and private sources.[137] Government investment in R&D increased 9.1 percent between 2022 and 2023 and was projected to increase by a further 6 percent in 2024.[138] Ireland’s research and innovation Strategy Impact 2030 outlines an ambition to increase R&D intensity to at least 2.5 percent by the end of 2030.[139] In 2023, Ireland’s R&D intensity was 1.59 percent.[140] For 2026, the government has earmarked €30 million of its €9.4 billion funding package to boost new, life-enhancing medicines.[141]
The Irish government encourages the development of the life sciences sector through an ecosystem of initiatives supporting manufacturing, innovation, and research. A foundational aspect of this system is the Industrial Development Authority (IDA), Ireland’s investment promotion agency responsible for attracting foreign direct investment. While active in more industries than just life sciences, IDA supports almost 1,800 companies to directly employ over 300,000 people in Ireland.[142] Another initiative, funded by IDA, is the National Institute for Bioprocessing Research and Training (NIRBT), a facility that convenes industry, government, and academia to support the development of biopharmaceutical manufacturing.[143]
The government also incentivizes innovation through tax policies. Companies engaged in R&D are eligible for a 30 percent tax credit on qualifying expenses.[144] Beginning on January 1, 2026, the rate was expected to increase to 35 percent.[145] Another tax policy promoting innovation is the Knowledge Development Box, a program providing corporate tax relief on income from IP created as a result of R&D activities.[146] This works as a complement to other innovation tax incentives. While the R&D tax credit provides support during the time firms conduct R&D, the Knowledge Development Box ensures future income generated from the results of R&D activity (i.e., IP) is partially protected from corporate tax.
Ireland placed 19th in the World Intellectual Property Organization’s (WIPO’s) Global Innovation Index 2024 report.[147] Businesses in Ireland can apply for IP protection through the Intellectual Property Office of Ireland, the European Union Intellectual Property Office, or the European Patent Office. Ireland is not yet fully participating in the Unified Patent Court (UPC) system, as ratification of the UPC Agreement is subject to the outcome of a pending referendum on this topic.[148]
The Netherlands
The Netherlands’ long-term goals for the country’s life sciences and biotechnology sectors are defined in the “Government’s Vision on Biotechnology 2025-2040,” an initiative that seeks to position the Netherlands as “a global leader in the research, development and application of biotechnology” by 2040.[149] Serving as a roadmap for ministerial collaboration and industry development, the document identifies several priority areas of societal impact to guide and accelerate biotechnological innovation. These include economic development, food security, and the circular economy. To achieve the government’s vision, the document calls for expanded research and innovation funding, a more proactive industrial policy, stronger IP rights, and increased investment in human capital. It also emphasizes the need for reduced strategic dependencies and more forward-looking regulation.
Life sciences and health is one of the Netherlands’ nine top sectors, targeted by the government to promote innovation and international recognition.[150] The country’s life sciences industry benefits from proximity to the EMA, located in Amsterdam, and is home to 420 biopharmaceutical firms.[151] The Netherlands’ total R&D expenditures in 2023 were €24.2 billion, with companies accounting for almost 60 percent and the government contributing roughly one-third of this amount.[152] As a share of GDP, the Netherlands’ R&D expenditures in 2023 amounted to approximately 2.27 percent. The government aims to increase this number to 3 percent by 2030, aligning itself with official EU targets.[153]
The Dutch government supports life sciences research through the National Growth Fund (NGF), which set aside €1.27 billion in 2025 to spur innovation in biotechnology.[154] The largest portion of funds is allocated to the BioBased Circular and Oncode Accelerator programs, aimed at developing climate-neutral materials and accelerating cancer therapies, respectively. Another notable project funded by the NGF is Biotech Booster, a program supporting the commercialization of biotech innovations.[155] Biotech Booster provides expert guidance and financial aid to promising ideas that fall into one of the following clusters: Industrial Biotechnology and Production, Agriculture and Food, Diagnostics and Services, Vaccines/Small Molecules, or Discovery and Development Platforms. Selected ideas receive up to €200,000 in the first two years, with the potential for up to €1.9 million in second-stage funding.[156] Biotech Booster recently joined forces with seven other NGF initiatives under the shared banner “Growing Together in Life Sciences & Health” to enhance collaboration across programs and strengthen innovation.[157]
Another important instrument for promoting life sciences is The Netherlands Organization for Health Research and Development (ZonMw), which provides funding for all stages of the innovation cycle, ranging from basic research to the deployment of new therapies.[158] The Netherlands also stimulates life sciences innovation through the research and development tax credit, WBSO, which provides both tax incentives and cash grants to companies and projects developing new products, production processes, or software.[159] Tax measures for eligible projects include lower labor costs and deductible research-related expenses. Companies that hold patents or otherwise profit from innovative creations can also qualify for the Innovation Box, which lowers corporate income tax for certain intangible assets.[160]
Sweden
Sweden’s national life sciences strategy calls for a “concerted effort for excellence, long-term competitiveness and increased patient benefit” and outlines an ambition “for Sweden to become a leading life sciences nation,” ensuring innovative treatments and interventions while simultaneously enhancing national competitiveness.[161] Updated in 2024, the strategy serves as a long-term framework for encouraging local, regional, and national mobilization toward eight priority areas. These include clinical trials, national digital infrastructure, medical technology and precision health, talent attraction, and international competitiveness. The strategy also emphasizes expanded opportunities for integrating research and innovation into health care and continued attention to emerging and strategically important technologies.
Sweden’s total intramural R&D expenditure in 2024 amounted to 230.4 billion SEK (approximately $22 billion).[162] This includes private sector investment, accounting for 74 percent of total expenditures.[163] Sweden’s government budget allocations for R&D reached 46.4 billion SEK ($4.4 billion) in 2024 and are expected to total 48.8 billion SEK ($5.1 billion) in 2025.[164] This is equal to 3.39 percent of the total government budget and represents an increase of around 5 percent compared with 2024. In 2023, Sweden’s R&D expenditure as a percentage of GDP was the fifth highest among OECD countries, at 3.57 percent.[165] The life sciences industry comprises 10 percent of Sweden’s total product exports.[166]
In 2024, the government earmarked 650 million SEK ($61.5 million) of its four-year 6.5 billion SEK ($615 million) research and innovation investment plan specifically for life sciences and health research.[167] Of this, 277.5 million SEK ($27 million) is set to go to Vinnova, Sweden’s innovation agency, tasked with developing an advanced life sciences program.[168] The effort shall be co-financed by the private sector, with industry contributing at least a corresponding share. The program seeks to promote cross-sectoral collaboration and expand research into strategic technologies, with the ultimate goal of strengthening Sweden’s attractiveness, creating new markets, and contributing to a more competitive and resilient ecosystem for the life sciences. As part of this program, Vinnova is also tasked with supporting the establishment of the Centre for Commercialization of Regenerative Medicine Nordic (CCRM Nordic)—a national innovation cluster supporting the commercialization and development of advanced therapies.[169] A further 360 million SEK ($34 million) of the total 650 million SEK ($61.5 million) life sciences budget is dedicated to boosting SciLifeLab, a national research center and cornerstone of Sweden’s effort to become a leading life sciences nation.[170]
PPPs are central to Sweden’s life sciences strategy and success. Examples include the Testa-Center, AstraZeneca BioVentureHub, and NorthX Innovation Hub.[171] These clusters enable innovation and commercialization by pooling resources and providing businesses with access to the facilities and infrastructure necessary for research and manufacturing.
Europe’s loss of biopharmaceutical leadership over the past several decades was not inevitable.
Tax deductions are available to companies engaging in R&D for commercial purposes with an emphasis on new knowledge creation. Deductions directly lower the monthly labor costs for personnel engaged in R&D activities by 20 percent.[172] Companies active in the life sciences space are also eligible to receive public or private grants. Since 2021, Vinnova has provided a total of 320 million SEK ($34.6 million) to 11 cross-border collaborations developing solutions across the entire health sector.[173] In 2024, Vinnova provided an additional 95 million SEK ($9 million) to finance six more projects, this time focusing on precision medicine.[174] The Knut and Alice Wallenberg Foundation also provides significant funding for life sciences research, having committed more than 6.8 billion SEK ($735 million) for the period 2014–2032.[175]
Sweden’s Patent Act was updated in 2025 to align with the European Patent Convention (EPC) and the Unified Patent Court (UPC).[176] Life sciences innovators can protect their products via the Swedish patent system, the EPC system, or through the supplemental UPC System. The Swedish Intellectual Property Office has developed several training modules in tandem with life sciences incubators to support early life sciences companies, start-ups, and entrepreneurs, helping them make informed decisions.[177] Sweden is one of the few countries to retain “professors’ privilege.”[178] This is the right of academics employed at Swedish higher education institutions to retain IP ownership of inventions developed in the workplace.
Another core tenet of Sweden’s life sciences strategy is international cooperation. In 2024, the government renewed its 2016 bilateral cooperation agreement with the United States to facilitate increased scientific exchange, promoting accelerated cancer research and care.[179] Following the renewal of the agreement, and the 2024 U.S.-Sweden Cancer Summit, the Swedish government tasked the National Board of Health and Welfare (Socialstyrelsen) with leading a joint steering committee responsible for the implementation of the agreement.[180] As part of this assignment, the National Board of Health and Welfare is expected to act as the convening authority for Swedish actors, submitting an interim report to the Ministry of Health and Social Affairs by March each year (Socialdepartementet). Although the Board was initially ordered to organize a 2025 U.S.-Sweden Cancer Summit in Sweden, this assignment has been postponed due to the current international research environment.[181]
United Kingdom
Already considered by many to be the European leader in biopharmaceuticals, the United Kingdom has announced plans to position itself as a top-three global hub for biopharmaceuticals by 2035. Already, the UK pharmaceutical industry contributes approximately £17.6 billion in direct gross value added annually to the British economy.[182] In January 2025, the UK government launched its comprehensive 10-year Life Sciences Sector Plan, which aims to advance innovation through aggressive investment in a multitude of facets, from R&D to manufacturing.[183] The strategy focuses primarily on overcoming post-Brexit challenges, strengthening manufacturing, and accelerating the adoption of innovation within the Medicines and Healthcare Products Regulatory Agency, the UK’s pharmaceutical regulatory bodies.
Currently, the United Kingdom leads the European region in biopharmaceuticals due to a combination of strong venture capital investment and the presence of major pharmaceutical players. The UK is the European region’s top biopharma VC market, capturing 30 percent of regional financing.[184] In 2025, the VC biopharmaceutical market in the UK was valued at £1.79 billion, and average deal sizes rose to £30.8 million.[185] These private investments provide significant footholds for a strong research innovation ecosystem. This system is bolstered by the strong presence of global firms, with several pharmaceutical companies, such as GSK and AstraZeneca, choosing to locate headquarters in city hubs like London and Cambridge. Other global companies, including Pfizer, Novartis, and Eli Lilly, also have a significant presence in the United Kingdom.[186] Finally, the UK is considered a hub for next-generation therapies, including AI-driven drug discovery and cell and gene therapy, and it hosts several start-ups in these areas, such as Autolus Therapies.[187]
The UK’s “patent box” regime represents a form of corporation tax relief designed to encourage companies to keep and commercialize IP. It rewards innovation by allowing companies to apply a lower rate of corporation tax to profits they earn from their patented inventions and certain other IP rights.[188]
However, despite the pharmaceutical sector emerging relatively unscathed from Brexit, performing comparatively stronger than other industries in the stock market post-referendum, the move has posed unique challenges to the industry.[189] Particularly, Brexit impacted the speed of innovation. Before Brexit, the UK was a part of the European Medicines Agency (EMA), which approved drugs for all of Europe. Afterwards, the UK’s Medicines and Healthcare products Regulatory Agency, once a part of the EMA, was forced to act independently for the first time, causing bottlenecks in the approval process. In 2021, the first year after Brexit, the UK approved 35 novel drugs, compared with 40 in the EU and 52 in the United States, and this pattern has continued for the past five years, with the UK trailing the rest of Europe by about five drugs per year.[190] Additionally, drugs often take comparatively longer to be approved in the UK, with the average regulatory approval lag between the UK and the EU being roughly one to two months.[191] This is because, after Brexit, companies must submit separate applications to the UK, and companies often submit to the EU first, since it affords access to a much larger market by population.
Thus, partly due to these post-Brexit challenges, the UK introduced the Life Sciences Sector Plan. According to the British government, “The Life Sciences Sector Plan outlines targeted actions to support world-class research and development, attract investment, grow advanced manufacturing, and accelerate health innovation.”[192] The plan aims to position the UK as the third global life sciences leader by 2035, behind only the United States and China. The report highlights the value of biopharmaceuticals by emphasizing that the sector is a leading driver of investment into the UK economy, and it made the largest contribution to business R&D performed in 2023, totaling 17 percent of all R&D performed by UK businesses.[193] The plan focuses on aggressive investment strategies to improve drug access times. The government pledged £520 million to the Life Sciences Innovative Manufacturing Fund (LSIMF) to bolster domestic production of medicines and diagnostics, and a record £86 billion for public R&D through 2030, with a focus on AI-driven drug discovery and clinical trial infrastructure.[194] Furthermore, the British government has sought to implement rules that would cut trial setup times to under 150 days by 2030, and established a goal of striking at least one major strategic partnership per year with leading biopharma firms.[195] Overall, the plan represents a holistic but intense approach to advancing the UK biopharmaceutical industry to global leadership by 2035.
Conclusion
Europe’s loss of biopharmaceutical leadership over the past several decades was not inevitable. It was the outcome of policies that weakened incentives for innovation, constrained investment, and slowed the translation of scientific advances into new therapies. While over this period the United States developed a supportive ecosystem that allowed the nation to grow into today’s global biopharmaceutical leader, growing U.S. interest in policies including drug price controls, reduced federal funding for basic scientific research, and weakened IP protections risks replicating some of the same policy missteps that contributed to Europe’s decline. As global competition intensifies—particularly from countries such as China—the United States cannot afford to undermine its biopharmaceutical innovation engine that has long served as a cornerstone of its economic strength, public health, and geopolitical influence.
To sustain leadership, U.S. policymakers should pursue strategies that reinforce the foundations of biopharmaceutical innovation, including strong and predictable IP rights, a gold-standard regulatory environment, the absence of government-imposed drug price controls, robust R&D investment, and a strong scientific workforce. Europe is now attempting to rebuild its competitiveness through substantial policy reforms and investment initiatives. The United States should view these efforts as a reminder that leadership in advanced technology industries can rapidly shift. Preserving America’s role as the world’s leading innovator in biopharmaceuticals will require it to recommit to a supportive policy environment that empowers scientific progress, rewards discovery, and keeps patients at the center of health innovation policy.
Acknowledgments
The authors would like to thank Robert D. Atkinson and Lila Trollip for helpful feedback on this report. Any errors or omissions are the authors’ sole responsibility.
About the Authors
Sandra Barbosu, Ph.D., is associate director of ITIF’s Center for Life Sciences Innovation. Her research focuses on the economics of health innovation, with a particular interest in the role of emerging technologies. She is also adjunct professor at New York University’s Tandon School of Engineering. She holds a Ph.D. in Strategic Management from the Rotman School of Management at the University of Toronto, an M.Sc. in Precision Cancer Medicine from the University of Oxford, and a B.A. in Economics and Mathematics from the University of Rochester.
Stephen Ezell is vice president for global innovation policy at ITIF and director of ITIF’s Center for Life Sciences Innovation. He also leads the Global Trade and Innovation Policy Alliance. His areas of expertise include science and technology policy, international competitiveness, trade, and manufacturing.
Esther Serger is a master’s candidate at Georgetown University’s School of Foreign Services.
About ITIF
The Information Technology and Innovation Foundation (ITIF) is an independent 501(c)(3) nonprofit, nonpartisan research and educational institute that has been recognized repeatedly as the world’s leading think tank for science and technology policy. Its mission is to formulate, evaluate, and promote policy solutions that accelerate innovation and boost productivity to spur growth, opportunity, and progress. For more information, visit itif.org/about.
Endnotes
[1]. Neil Turner, “What’s gone wrong with the European pharmaceutical industry,” The Pharma Letter, April 29, 1999, https://www.thepharmaletter.com/what-s-gone-wrong-with-the-european-pharmaceutical-industry-by-neil-turner; David Michels and Aimison Jonnard, “Review of Global Competitiveness in the Pharmaceutical Industry” (US. International Trade Commission, 1999), 2–3, https://wwwusitc.gov/publications/332/pub3172.pdf.
[2]. John K Jenkins, MD., “CDER New Drug Review: 2015 Update” (PowerPoint presentation, US Food and Drug Administration/CMS Summit, Washington, D.C., December 14, 2015), http://wwwfda.gov/downloads/AboutFDA/CentersOffices/OfficeofMedicalProductsandTobacco/CDER/UCM477020.pdf.
[3]. European Federation of Pharmaceutical Industries and Associations (EFPIA), “The Research Based Pharmaceutical Industry: A Key Actor for a Healthy Europe,” (EFPIA, 2006).
[4]. Tom Randall, “Even European’s don’t see a future in Euro biopharma anymore,” Endpoint News, February 12, 2026, https://endpoints.news/even-europeans-dont-see-a-future-in-euro-biopharma-anymore/.
[5]. Kimberly Cleaves, “Imbalanced Innovation,” Modern Drug Discovery (July 2004): 23–24, http://pubsapp.acs.org/subscribe/archive/mdd/v07/i07/pdf/704business3.pdf.
[6]. Nathalie Moll, “Would the last pharmaceutical investor in Europe please turn the lights out,” European Federation of Pharmaceutical Industries and Associations, January 3, 2020, https://www.efpia.eu/news-events/the-efpia-view/blog-articles/would-the-last-pharmaceutical-investor-in-europe-please-turn-the-lights-out/.
[7]. Arthur Daemmrich, “Where is the Pharmacy to the World? International Regulatory Variation and Pharmaceutical Industry Location,” (Harvard Business School, Working Paper 09-11, 2009), www.hbs.edu/ris/Publication%20Files/09-118.pdf.
[8]. Moll, “Would the last pharmaceutical investor in Europe please turn the lights out.”
[9]. Neil Turner, “European Pricing Squeeze,” PharmaExeccom, October 1, 2002, https://www.pharmexec.com/view/european-pricing-squeeze.
[10]. Lakdawalla et al., “U.S. Pharmaceutical Policy In A Global Marketplace” (January 2008), https://www.rand.org/content/dam/rand/pubs/reprints/2009/RAND_RP1380.pdf; Turner, “European Pricing Squeeze”; David J. Gross, “International Pharmaceutical Spending Controls: France, Germany, Sweden, and the United Kingdom,” Health Care Financing Review Vol. 15, No. 3 (1994): 27–140, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4193451/.
[11]. Giovanni Giuliani, Gisbert Selke, and Livio Garattini, “The German experience in reference pricing,” Health Policy, Vol. 44, No. 1 (1998): 73–85, https://www.sciencedirect.com/science/article/abs/pii/S0168851098000128.
[12]. U.S. Government Accountability Office (GAO) “Prescription Drugs: Spending Controls in Four European Countries,” (GAO, May 1994), 56, https://www.gao.gov/assets/hehs-94-30.pdf.
[13]. Giuliani, Selke, and Garattini, “The German experience in reference pricing.”
[14]. Livio Garattini, Dante Cornago, and Paola De Compadri. “Pricing and reimbursement of in-patent drugs in seven European countries: a comparative analysis,” Health Policy, Vol. 82, No. 3 (2007): 330–339, https://pubmed.ncbi.nlm.nih.gov/17125881/.
[15]. “Keeping medicines affordable,” Government of the Netherlands, accessed February 12, 2026, https://www.government.nl/topics/medicines/keeping-medicines-affordable.
[16]. GAO, “Prescription Drugs: Spending Controls in Four European Countries.”
[17]. Veronique C. Raimond et al., “Why France spends less than the United States on drugs: a comparative study of drug pricing and pricing regulation,” The Milbank Quarterly, Vol. 99, No. 1 (2021): 240–272, https://pmc.ncbi.nlm.nih.gov/articles/PMC7984670/.
[18]. GAO, “Prescription Drugs: Spending Controls in Four European Countries.”
[19]. Cleaves, “Imbalanced Innovation,” 23
[20]. Joseph H. Golec and John A. Vernon, “European Pharmaceutical Price Regulation, Firm Profitability, and R&D Spending” (National Bureau of Economic Research Working Paper 12676, November 2006), 6–7, https://www.nber.org/system/files/working_papers/w12676/w12676.pdf.
[21]. Andrew S. Mulcahy, Daniel Schwam, and Susan L. Lovejoy, “International Prescription Drug Price Comparisons, Estimates using 2022 Data,” (RAND Corporation, February 2024), https://www.rand.org/pubs/research_reports/RRA788-3.html.
[22]. Ibid.
[23]. Ibid.
[24]. Jim Gilbert and Paul Rosenberg, “Addressing the Innovation Divide: Imbalanced innovation” (Bain & Company, 2004), https://www.bain.com/insights/addressing-the-innovation-divide-imbalanced-innovation/.
[25]. Doug Badger, “Examination of International Drug Pricing Policies in Selected Countries Shows Prevalent Government Control over Pricing and Restrictions on Access,” (Galen Institute, March 2019), https://galen.org/assets/Badger-Report-March-2019.pdf.
[26]. IQVIA, “Assessing Availability of New Drugs in Europe, Japan, and the U.S.,” December 11, 2024, https://www.iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/assessing-availability-of-new-drugs-in-europe-japan-and-the-us.
[27]. Lakdawalla et al., “U.S. Pharmaceutical Policy In A Global Marketplace,” 3.
[28]. Ibid, 9.
[29]. “United States Population by Age,” Neilsberg, February 22, 2025, https://www.neilsberg.com/insights/united-states-population-by-age/.
[30]. Joseph Golec and John A. Vernon, “Financial effects of pharmaceutical price regulation on R&D spending by EU versus US firms,” Pharmacoeconomics Vol. 28 (2010): 615–628, https://pubmed.ncbi.nlm.nih.gov/20617857/.
[31]. European Federation of Pharmaceutical Industries and Associations (EFPIA), “The Pharmaceutical Industry in Figures, Key Data 2024,” 8, https://www.efpia.eu/media/2rxdkn43/the-pharmaceutical-industry-in-figures-2024.pdf.
[32]. Ibid.
[33]. Golec and Vernon, “European Pharmaceutical Price Regulation, Firm Profitability, and R&D Spending,” 3
[34]. Ibid, 28.
[35]. CA. Brouwers, M.B. Silverstein, and T. Wolff, “Adverse Consequences of OECD Government Interventions in Pharmaceutical Markets on the U.S. Economy and Consumer” (The Boston Consulting Group Inc., 2004).
[36]. Kate Ho and Ariel Pakes, “Policy options for the drug pricing conundrum,” Proceedings of the National Academy of Sciences Vol. 122, No. 9 (2024), https://www.pnas.org/doi/10.1073/pnas.2418540122; Trelysa Long and Stephen Ezell, “The Hidden Toll of Drug Price Controls: Fewer New Treatments and Higher Medical Costs for the World” (ITIF, July 2023), https://itif.org/publications/2023/07/17/hidden-toll-of-drug-price-controls-fewer-new-treatments-higher-medical-costs-for-world/.
[37]. Ho and Pakes, “Policy options for the drug pricing conundrum.”
[38]. EUR-Lex, “The precautionary principle,” https://eur-lex.europa.eu/EN/legal-content/summary/the-precautionary-principle.html.
[39]. Daemmrich, “Where is the Pharmacy to the World?” 10.
[40]. Aurelien Portuese, “Pharmaceutical Consolidation & Competition: A Prescription for Innovation” (ITIF, June 2021), https://www2.itif.org/2021-pharmaceutical-task-force.pdf.
[41]. McKinsey & Company, “Europe’s start-up ecosystem: Heating up, but still facing challenges,” October 11, 2020, https://www.mckinsey.com/industries/technology-media-and-telecommunications/our-insights/europes-start-up-ecosystem-heating-up-but-still-facing-challenges.
[42]. Daemmrich, “Where is the Pharmacy to the World?” 1.
[43]. Ibid, 10.
[44]. Joe Kennedy, “The Link Between Drug Prices and Research on the Next Generation of Cures” (ITIF, September 2019), https://itif.org/publications/2019/09/09/link-between-drug-prices-and-research-next-generation-cures/.
[45]. Destrina Grace Simanjuntak and Raymond R. Tjandrawinata, “Impact of Profitability, R&D Intensity, and Cash Flow on R&D Expenditure in Pharmaceutical Companies” (Working paper, Social Science Research Network, April 29, 2011), http://papers.ssrn.com/sol3/papers.cfm?abstract_id=1824267.
[46]. Alfonso Gambardella, Science and Innovation: The US Pharmaceutical Industry During the 1980s (Cambridge University Press, 1995).
[47]. Rebecca Henderson and Iain Cockburn, “Measuring Competence? Exploring Firm Effects in Pharmaceutical Research,” Strategic Management Journal Vol. 15, No. S1 (1994): 63–84, https://doi.org/10.1002/smj.4250150906.
[48]. Pierre Dubois, “Market size and pharmaceutical innovation,” The Rand Journal of Economics Vol. 46, No. 4 (October 2015): 844–871, https://onlinelibrary.wiley.com/doi/full/10.1111/1756-2171.12113.
[49]. Golec and Vernon, “Financial effects of pharmaceutical price regulation on R&D spending by EU versus US firms.”
[50]. Lakdawalla et al., “U.S. Pharmaceutical Policy In A Global Marketplace.”
[51]. Congressional Research Service (CRS), “National Institutes of Health (NIH) Funding: FY1996–FY2023,” (CRS, March 8, 2023), https://crsreports.congress.gov/product/pdf/R/R43341/45; National Institutes of Health, “Budget,” accessed January 11, 2024, https://www.nih.gov/about-nih/what-we-do/budget; National Institutes of Health, “Mission and Goals,” https://www.nih.gov/about-nih/what-we-do/mission-goals.
[52]. Johns Hopkins University, “5 things to know about the power of mRNA technology,” April 15, 2025, https://washingtondc.jhu.edu/news/5-things-to-know-about-mrna/; DARPA, “Adept/P3,” June 2, 2017, https://www.darpa.mil/node/3710; DARPA, “Biological Technologies Office,” https://www.darpa.mil/about/offices/bto.
[53]. ARPA-H, “About Us,” https://arpa-h.gov/about; ARPA-H, “Budget and Appropriations,” https://arpa-h.gov/about/budget.
[54]. Stephen Ezell, Meghan Ostertag, and Leah Kann, “The Bayh-Dole Act’s Role in Stimulating University-Led Regional Economic Growth” (ITIF, June 2025), https://itif.org/publications/2025/06/16/bayh-dole-acts-role-in-stimulating-university-led-regional-economic-growth/.
[55]. Ashish Arora et al., “The changing structure of American innovation: Some cautionary remarks for economic growth,” Innovation Policy and the Economy Vol. 20, No. 1 (2020): 39–93, https://www.journals.uchicago.edu/doi/full/10.1086/705638.
[56]. Ibid.
[57]. Ashish Arora, Sharon Belenzon, and Andrea Patacconi, “Papers to patents,” Nature Vol. 552, No. 7683 (2017): S10, https://www.nature.com/articles/d41586-017-07421-3.
[58]. Ezell, Ostertag, and Kann, “The Bayh-Dole Act’s Role in Stimulating University-Led Regional Economic Growth.”
[59]. John Swann, “The Story Behind the Orphan Drug Act,” (FDA, February 23, 2018), https://www.fda.gov/industry/fdas-rare-disease-day/story-behind-orphan-drug-act.
[60]. Lewis J. Fermaglich and Kathleen L. Miller, “A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act,” Orphanet Journal of Rare Diseases Vol. 18 (2023), 163, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10290406/.
[61]. Ernst R. Berndt et al., “Assessing the impacts of the prescription drug user fee acts (PDUFA) on the FDA approval process,” Forum for Health Economics & Policy Vol. 8, No. 1. (2005), https://doi.org/10.2202/1558-9544.1013.
[62]. Amitabh Chandra et al., “Regulatory Incentives for Innovation: The FDA’s Breakthrough Therapy Designation,” The Review of Economics and Statistics (2026), 1–15.
[63]. Bacchus Barua, Will Westcott, and Van Nghiem Vo, “Timely Access to New Pharmaceuticals in Canada, the United States, and the European Union” (Fraser Institute, 2021), https://www.fraserinstitute.org/sites/default/files/timely-access-to-new-pharmaceuticals-in-canada-US-and-EU.pdf.
[64]. David Proudman et al., “Public sector replacement of privately funded pharmaceutical R&D: cost and efficiency considerations,” Journal of Medical Economics Vol. 27, No. 1 (2024): 125301266.
[65]. Helen Dowden and Jamie Munro, “Trends in clinical success rates and therapeutic focus,” Nature, From The Analyst’s Couch, Vol. 18 (2019): 495–496.
[66]. Stephen Ezell and Kelli Zhao, “The Economics of Biopharmaceutical Innovation: Symposium Report” (ITIF, March 2023), https://itif.org/publications/2023/03/20/economics-of-biopharmaceutical-innovation-symposium-report/.
[67]. Heidi L. Williams, “How do patents affect research investments?” Annual Review of Economics Vol. 9 (2017): 441–469, https://www.annualreviews.org/doi/10.1146/annurev-economics-110216-100959.
[68]. Olga Gurgula, “Strategic patenting by pharmaceutical companies–should competition law intervene?” IIC-International Review of Intellectual Property and Competition Law Vol. 51, No. 9 (2020): 1062–1085, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7592140/.
[69]. Ashish Arora, Marco Ceccagnoli, and Wesley M. Cohen, “R&D and the patent premium,” International Journal of Industrial Organization Vol. 26, No. 5 (2008): 1153–1179, https://www.sciencedirect.com/science/article/abs/pii/S0167718707001373.
[70]. Margaret Kyle and Yi Qian, “Intellectual property rights and access to innovation: evidence from TRIPS” (Working paper, National Bureau of Economic Research, 2014), https://www.nber.org/papers/w20799.
[71]. Iain M. Cockburn et al., “Patents and the global diffusion of new drugs,” American Economic Review Vol. 106, No. 1 (2016): 136–164, https://www.aeaweb.org/articles?id=10.1257/aer.20141482.
[72]. John P. Walsh, Ashish Arora, and Wesley M. Cohen, “Effects of research tool patents and licensing on biomedical innovation,” Patents in the Knowledge-based Economy Vol. 285 (2003): 286, http://elibrary.pcu.edu.ph:9000/digi/NA02/2003/10770.pdf#page=298.
[73]. Dana P. Goldman, “Regulating Drug Prices: U.S. Policy Alternatives in a Global Context,” RAND Corporation (2008), https://www.rand.org/pubs/research_briefs/RB9412.html.
[74]. IQVIA, “Drug Expenditure Dynamics 1995–2020: Understanding Medicine Spending in Context,” Institute for Human Data Science (2021), https://www.iqvia.com/-/media/iqvia/pdfs/institute-reports/drug-expenditure-dynamics/drug-expenditure-dynamics-19952020.pdf.
[75]. Stephen Ezell and Kelli Zhao, “How Skeptics Misconstrue the Link Between Drug Prices and Innovation” (ITIF, September 2023), https://itif.org/publications/2023/09/05/how-skeptics-misconstrue-the-link-between-drug-prices-and-innovation/.
[76]. Bureau of Labor Statistics, “Consumer Expenditure Survey (Healthcare expenditures, 2009–2024),” accessed February 11, 2026, https://www.bls.gov/cex/.
[77]. Ibid.
[78]. Centers for Medicare & Medicaid Services, “Medicare Drug Price Negotiation Program: Selected Drugs for Initial Price Applicability Year 2026,” Medicare Drug Price Negotiation Program (August 2023), https://www.cms.gov/files/document/fact-sheet-medicare-selected-drug-negotiation-list-ipay-2026.pdf.
[79]. Duane Schulthess et al., “The Inflation Reduction Act’s Impact upon Early-Stage Venture Capital Investments,” Therapeutic Innovation & Regulatory Science (2025), https://www.medrxiv.org/content/10.1101/2025.01.07.25320113v1.
[80]. PhRMA, “Inflation Reduction Act Already Impacting R&D,” https://cdn.aglty.io/phrma/global/resources/import/pdfs/Infographic%20%20Inflation%20Reduction%20Act%20Already%20Impacting%20RD%20%20010923%20FINAL.pdf.
[81]. Vital Transformation, “Inflation Reduction Act – Two Years On; Investor Behavior, R&D Impacts, & Proposed Solutions,” https://vitaltransformation.com/2025/04/inflation-reduction-act-two-years-on-investor-behavior-rd-impacts-proposed-solutions/.
[82]. Tomas J. Philipson and Troy Durie, “The evidence base on the impact of price controls on medical innovation” (Working paper, Becker Friedman Institute, University of Chicago, 2021), https://bfi.uchicago.edu/working-paper/the-evidence-base-on-the-impact-of-price-controls-on-medical-innovation/.
[83]. U.S. Chamber of Commerce Global Innovation Policy Center “From Innovation Oasis to Research Desert: The Impact of Price Controls on Clinical Research and Development,” (U.S. Chamber of Commerce, 2023), https://www.uschamber.com/assets/documents/From-Innovation-Oasis-to-Research-Desert-USCC.pdf.
[84]. The White House, “Delivering most-favored-nation prescription drug pricing to American patients,” May 12, 2025, https://www.whitehouse.gov/presidential-actions/2025/05/delivering-most-favored-nation-prescription-drug-pricing-to-american-patients/.
[85]. Stephen Ezell, “Comments to Department of Health and Human Services Regarding the Proposed GUARD Model” (ITIF, February 23, 2026), https://itif.org/publications/2026/02/23/comments-department-health-human-services-regarding-proposed-guard-model/; Stephen Ezell, “Comments to Department of Health and Human Services Regarding the Proposed GLOBE Model,” (ITIF, February 23, 2026), https://itif.org/publications/2026/02/23/comments-department-health-human-services-regarding-proposed-globe-model/.
[86]. Sandra Barbosu, “How Innovative is China in Biotechnology?” (ITIF, July 2024), https://itif.org/publications/2024/07/30/how-innovative-is-china-in-biotechnology/.
[87]. Stephen Ezell, “Comments to the Bureau of Industry and Security Regarding Its Section 232 Investigation of Pharmaceutical Imports” (ITIF, May 7, 2025), https://itif.org/publications/2025/05/07/comments-regarding-section-232-investigation-of-pharmaceutical-imports/.
[88]. Ibid.
[89]. Richard G. Frank, “The 2026 health and health care budget” (Brookings Institution, June 2025), https://www.brookings.edu/articles/the-2026-health-and-health-care-budget/.
[90]. Andrew Matthius, “Senate Forum Examined the Ramifications of NIH Funding Cuts,” Cancer Research Catalyst (AACR blog), April 16, 2025,https://www.aacr.org/blog/2025/04/16/senate-forum-examined-the-ramifications-of-nih-funding-cuts/.
[91]. Kritika Agarwal, “NIH Funding Cuts Will Result in Fewer Drugs Coming to Market, CBO Finds,” Association of American Universities, July 25, 2025, https://www.aau.edu/newsroom/leading-research-universities-report/nih-funding-cuts-will-result-fewer-drugs-coming.
[92]. Arora et al., “The changing structure of American innovation.”
[93]. Ibid.
[94]. Birch Bayh, “Statement of Birch Bayh to the National Institutes of Health,” May 25, 2014, http://www.essentialinventions.org/drug/nih05252004/birchbayh.pdf.
[95]. Joseph Allen, “Proposal from Senator King Won’t Reduce Drug Prices, Just Innovation,”IPWatchdog, July 17, 2017, https://www.ipwatchdog.com/2017/07/17/senator-king-reduce-drug-prices-innovation/id=85702/.
[96]. Stephen Ezell, “Comments to the National Institutes of Health on ‘Maximizing NIH’s Levers to Catalyze Technology Transfer’” (ITIF, August 2023), https://itif.org/publications/2023/08/18/maximizing-nih-levers-to-catalyze-technology-transfer/.
[97]. National Institute of Standards and Technology (NIST) “Return on Investment Initiative: Draft Green Paper” (NIST, December 2018), 30, https://nvlpubs.nist.gov/nistpubs/SpecialPublications/NIST.SP.1234.pdf.
[98]. Ibid.
[99]. Rachel Cohrs Zhang, “RFK Jr. says U.S. won’t threaten pharmaceutical patents to push for lower drug prices,” STAT+, January 31, 2025, https://www.statnews.com/2025/01/31/rfk-jr-trump-hhs-pick-kennedy-march-in-rights-drug-prices/.
[100]. National Security Commission on Emerging Biotechnology (NSCEB), “Charting the Future of Biotechnology,” (NSCEB, April 2025), https://www.biotech.senate.gov/final-report/chapters/.
[101]. European Commission, “Competitiveness compass” (Brussels: European Commission, 2025), https://commission.europa.eu/topics/competitiveness/competitiveness-compass_en.
[102]. Directorate-General for Communication, “Making Europe a global leader in life sciences,” news release, July 2, 2025, https://commission.europa.eu/news-and-media/news/making-europe-global-leader-life-sciences-2025-07-02_en.
[103]. Thomas Gabrielczyk, “EU set to improve investment for biotech start-ups and scale-ups,” European Biotechnology Magazine, May 30, 2025, https://european-biotechnology.com/latest-news/eu-set-to-improve-investment-for-biotech-start-ups-and-scale-ups/.
[104]. “Choose Europe,” The European Commission, accessed December 2, 2025, https://commission.europa.eu/topics/research-and-innovation/choose-europe_en.
[105]. Directorate-General for Communication, “Making Europe a global leader in life sciences.”
[106]. European Commission, “EIC Impact Report 2023: Accelerating Deep Tech in Europe,” news release, March 25, 2024, https://ec.europa.eu/newsroom/eismea/items/824162/en.
[107]. European Commission, “Commission launches new strategy to make Europe a global leader in life sciences by 2030,” press release, July 1, 2025, https://ec.europa.eu/commission/presscorner/detail/en/ip_25_1686.
[108]. “Public-private partnership for bio-based industries,” European Union, last modified May 16, 2018, https://eur-lex.europa.eu/EN/legal-content/summary/public-private-partnership-for-bio-based-industries.html.
[109]. Hildegard Bentele, “DRAFT REPORT: on the future of the EU biotechnology and biomanufacturing sector: leveraging research, boosting innovation and enhancing competitiveness” (Brussels: European Parliament Committee on Industry, Research and Energy, March 2025), https://www.europarl.europa.eu/doceo/document/ITRE-PR-768143_EN.pdf.
[110]. European Medicines Agency (EMA), “EMA welcomes political agreement on new EU pharmaceutical legislation,” EMA, December 2025, https://www.ema.europa.eu/en/news/ema-welcomes-political-agreement-new-eu-pharmaceutical-legislation; “Streamlining change: New EU guidelines simplify medicines lifecycle management,” GMDP Academy, October 2025, https://gmdpacademy.org/news/streamlining-change-new-eu-guidelines-simplify-medicines-lifecycle-management/.
[111]. “Future Research and Innovation Strategy” (Berlin: The Federal Government, 2023), https://www.aufstiegs-bafoeg.de/SharedDocs/Publikationen/de/bmbf/FS/747580_Zukunftsstrategie_Forschung_und_Innovation_en.pdf.
[112]. “European reference pricing is discontinued in Germany,” Value & Dossier, https://value-dossier.com/en/european-reference-pricing-is-discontinued-in-germany/.
[113]. “National Bioeconomy Strategy” (Berlin: The Federal Government, July 2020), https://www.bmftr.bund.de/SharedDocs/Publikationen/DE/FS/31617_Nationale_Biooekonomiestrategie_Langfassung_en.pdf.
[114]. “GO-Bio - Seal of quality in the life sciences,” Federal Ministry of Research, Technology, and Space, accessed November 18, 2025, https://www.go-bio.de/gobio/en/go-bio/go-bio_node.html.
[115]. Federal Ministry of Research, Technology, and Space, “Federal Report on Research and Innovation 2024” (Berlin: Federal Ministry of Research, Technology, and Space, May 2024), 4, https://www.bundesbericht-forschung-innovation.de/files/BMBF_BuFI-2024_Short-version.pdf.
[116]. Dresden University of Technology, “From Lab to Business: CRTD Researchers receive BMBF GO-Bio Initial Funding,” news release, October 26, 2022, https://tu-dresden.de/cmcb/crtd/news-termine/news/from-lab-to-business-crtd-researchers-receive-bmbf-go-bio-funding?set_language=en; German Federal Ministry of Research, Technology, and Space, “The Cluster Initiative,” accessed November 18, 2025, https://www.clusters4future.de/homepage.
[117]. Federal Ministry of Education and Research, “Bioeconomy International” (Berlin: Federal Ministry of Education and Research, September 2024), 5, https://www.bmftr.bund.de/SharedDocs/Publikationen/DE/FS/1060324_Biooekonomie_International_en.pdf.
[118]. “Munich Biotech Cluster m4,” Clusterplattform Deutschland, accessed November 18, 2025, https://www.clusterplattform.de/CLUSTER/Redaktion/EN/Cluster_Archiv/go-cluster/munich_biotech_cluster_m4.html.
[119]. “BMBF Initiative “German Centers for Health Research,” Deutsches Zentrum für Herz-Kreizlauf-Forschung, accessed November 18, 2025, https://dzhk.de/en/the-dzhk/about-us/bmbf-initiative.
[120]. “R&D tax credit (Forschungszulage),” The OECD INNOTAX portal, accessed November 18, 2025, https://stip.oecd.org/innotax/incentives/DEU1.
[121]. EY Global, “German Federal Council approves amended Growth Opportunities Act bill on corporate tax reform,” news alert, March 25, 2024, https://www.ey.com/en_gl/technical/tax-alerts/german-federal-council-approves-amended-growth-opportunities-act.
[122]. European Patent Office, “When was the Unitary Patent system launched?” accessed November 18, 2025, https://www.epo.org/en/applying/european/unitary/unitary-patent/start.
[123]. German Patent and Trademark Office, “Utility Models,” last modified October 20, 2020, https://www.dpma.de/english/utility_models/index.html.
[124]. German Patent and Trademark Office, “Examination Guidelines for Supplementary Protection Certificates” (Berlin: German Patent and Trade Mark Office, January 2015), https://www.dpma.de/docs/english/formulare/patent_eng/p2799_1.pdf.
[125]. Government of France, “Healthcare innovation 2030,” (Paris: Government of France, June 2021), https://www.enseignementsup-recherche.gouv.fr/sites/default/files/content_migration/document/sante_innov30_A4_07_-_ENG_1415002.pdf.
[126]. Government of France, “Une stratégie pour une France leader dans la production de thérapies innovantes,” news release, January 10, 2022, https://www.economie.gouv.fr/strategie-france-leader-production-therapies-innovantes.
[127]. Government of France, “Healthcare innovation 2030,” 5–7.
[128]. Health Innovation Agency, “ROADMAP: for the Health Innovation Agency” (Paris: Government of France, April 2024), https://www.info.gouv.fr/upload/media/content/0001/10/440a92a17aa78c3b5585acaf780189a16fb394d2.pdf.
[129]. Government of France, “Choose France for Science: launch of the dedicated platform for applications to host international researchers,” news release, April 17, 2025, https://anr.fr/en/latest-news/read/news/choose-france-for-science-launch-of-the-dedicated-platform-for-applications-to-host-international-r/.
[130]. “Tax incentives,” Government of France, accessed December 2, 2025, https://www.impots.gouv.fr/international-professionnel/tax-incentives; “R&D tax credit – Crédit d’Impôt Recherche (CIR),” The OECD INNOTAX portal, accessed December 2, 2025, https://stip.oecd.org/innotax/incentives/FRA1.
[131]. “Les jeunes entreprises innovantes (JEI),” Urssaf, last modified March 14, 2025, https://www.urssaf.fr/accueil/employeur/beneficier-exonerations/exonerations-secteur-activite/jeunes-entreprises-innovantes.html.
[132]. “Extending your patent abroad,” French National Institute of Industrial Property, accessed December 2, 2025, https://www.inpi.fr/en/realiser-demarches/propriete-intellectuelle/etendre-son-brevet-letranger.
[133]. Programme for Government: Securing Ireland’s Future (Dublin: Ireland Department of the Taoiseach, January 2025), https://www.gov.ie/en/department-of-the-taoiseach/publications/programme-for-government-2025-securing-irelands-future/.
[134]. “Ireland: at the heart of global health & life sciences supply chains” (KPMG, November 2024), https://kpmg.com/ie/en/insights/healthcare/ireland-global-health-life sciences-supply-chain.html; “Ireland: How The Irish Life Sciences Sector is Leading the World” (NSF, October 2024), https://www.nsf.org/knowledge-library/ireland-life-sciences-overview.
[135]. IDA Ireland, “Ireland: A Global Center for Life Sciences,” news insights, June 28, 2023, https://www.idaireland.com/latest-news/insights/ireland-a-global-center-for-life-sciences.
[136]. IDA Ireland, “Key players in Ireland’s pharmaceutical manufacturing space,” news insights, July 9, 2025, https://www.idaireland.com/latest-news/insights/pharmaceutical-manufacturing-companies-ireland.
[137]. The Research and Development Budget 2023 to 2024 (Dublin: Ireland Department of Further and Higher Education, Research, Innovation and Science, September 2025), https://www.gov.ie/en/department-of-further-and-higher-education-research-innovation-and-science/publications/the-research-and-development-budget-2023-to-2024/.
[138]. Ibid., 18–19.
[139]. Impact 2030: Ireland’s Research and Innovation Strategy (Dublin: Ireland Department of Further and Higher Education, Research, Innovation and Science, May 2022), https://assets.gov.ie/static/documents/impact-2030-irelands-research-and-innovation-strategy.pdf.
[140]. The Research and Development Budget 2023 to 2024, 39.
[141]. Brian Maguire, “Irish pharma sector hails €30m medicines boost and R&D tax credit increase,” Euractiv, October 9, 2025, https://www.euractiv.com/news/irish-pharma-sector-hails-e30m-medicines-boost-and-rd-tax-credit-increase/.
[142]. “About IDA Ireland,” IDA Ireland, accessed November 13, 2025, https://www.idaireland.com/about-ida-ireland.
[143]. “About NIBRT,” NIBRT, accessed November 13, 2025, https://www.nibrt.ie/about/.
[144]. “Research and Development (R&D) Corporation Tax credit,” Revenue: Irish Tax and Customs, accessed November 13, 2025, https://www.revenue.ie/en/companies-and-charities/reliefs-and-exemptions/research-and-development-rd-tax-credit/index.aspx.
[145]. Damien Flanagan, “R&D incentives - Budget 2026” (KPMG, October 2025), https://kpmg.com/ie/en/insights/tax/budget-2026/rd-incentives.html.
[146]. “Knowledge Development Box (KDB),” Revenue: Irish Tax and Customs, accessed November 13, 2025, https://www.revenue.ie/en/companies-and-charities/reliefs-and-exemptions/knowledge-development-box-kdb/index.aspx.
[147]. World Intellectual Property Organization (WIPO), “Global Innovation Index 2024: Unlocking the Promise of Social Entrepreneurship” (Geneva: WIPO, 2024), 18, https://www.wipo.int/web-publications/global-innovation-index-2024/assets/67729/2000%20Global%20Innovation%20Index%202024_WEB3lite.pdf.
[148]. Pinsent Mansons, “Renewed interest in Irish UPC referendum welcomed,” news release, August 19, 2025, https://www.pinsentmasons.com/out-law/news/renewed-interest-irish-upc-referendum.
[149]. “Government’s Vision on Biotechnology 2025-2040” (Amsterdam: Government of the Netherlands, May 2025), https://www.government.nl/documents/reports/2025/04/11/dutch-vision-on-biotechnology-2025-2040.
[150]. “Encouraging innovation,” Government of the Netherlands, accessed February 9, 2026, https://www.government.nl/topics/enterprise-and-innovation/encouraging-innovation.
[151]. “Life Sciences & Health,” Netherlands, accessed February 9, 2026, https://www.welcome-to-nl.nl/work-in-nl/life-sciences-health.
[152]. “Funding and performance of R&D in the Netherlands,” Rathenau Instituut, accessed January 7, 2026, https://www.rathenau.nl/en/science-figures/investments/how-much-does-netherlands-spend-rd/funding-and-performance-rd-netherlands.
[153]. Government of the Netherlands, “Government’s Vision on Biotechnology 2025-2040,” 30.
[154]. Ibid., 31.
[155]. “About us,” Biotech Booster, accessed January 9, 2026, https://www.biotechbooster.nl/about-us/.
[156]. Ibid.
[157]. PharmaNL, “Eight National Growth Fund initiatives join forces to strengthen the Dutch Life Sciences & Health ecosystem,” news release, December 4, 2025, https://www.pharmanl.org/en/news/eight-national-growth-fund-initiatives-join-forces/.
[158]. “About ZonMw,” accessed February 9, 2026, https://www.zonmw.nl/en/about-zonmw.
[159]. Deloitte, “Grants and Incentives for the Life Sciences & Healthcare industry,” accessed February 9, 2026, https://www.deloitte.com/nl/en/services/tax/services/grants-and-incentives-in-life-sciences-healthcare-industry.html.
[160]. Netherlands Enterprise Agency, “Innovation box,” accessed February 9, 2026, https://business.gov.nl/subsidies-and-schemes/innovation-box/.
[161]. A concerted effort for long-term excellence, competitiveness and increased patient benefit, Stockholm: Government Offices of Sweden, February 2025, https://www.government.se/information-material/2025/02/a-concerted-effort-for-excellence-long-term-competitiveness-and-increased-patient-benefit/.
[162]. Statistics Sweden, “No increase in R&D expenditure during 2024,” statistical news, July 10, 2025, https://www.scb.se/en/finding-statistics/statistics-by-subject-area/research-and-the-digital-society/research-and-development/research-and-development-in-sweden/pong/statistical-news/research-and-development-in-sweden-2024---preliminary-statistics/.
[163]. Ibid.
[164]. Statistics Sweden, Government budget allocations for R&D (SEK million by year, accessed November 13, 2025), https://www.statistikdatabasen.scb.se/pxweb/en/ssd/START__UF__UF0306__UF0306A/SBAUtg/; Statistics Sweden, “Government budget allocations for R&D 2025,” statistical news, March 20, 2025, https://www.scb.se/en/finding-statistics/statistics-by-subject-area/research-and-the-digital-society/research-and-development/government-budget-allocations-for-rd/pong/statistical-news/government-budget-allocations-for-rd-2025--preliminary-data/.
[165]. Sarah Coats Chandler, “Life Science Overview” (Stockholm: The Swedish Institute, 2025), https://www.business-sweden.com/496e69/globalassets/insights/reports/invest/si_ip_life-science_overview.pdf.
[166]. “Life Science Barometer 2025” (SwedenBIO, December 2024), https://www.mediconvillage.se/app/uploads/2024/12/the-life-science-barometer-2025-lowres.pdf.
[167]. Government Offices of Sweden, “Excellent research and innovation are encouraged in largest-ever research and innovation bill,” press release, December 16, 2024, https://www.government.se/press-releases/2024/12/excellent-research-and-innovation-are-encouraged-in-largest-ever-research-and-innovation-bill/.
[168]. Government Offices of Sweden, “Uppdrag till Verket för innovationssystem att inrätta ett program för avancerad life science” (Stockholm: Government Offices of Sweden, July 2025), https://www.regeringen.se/regeringsuppdrag/2025/07/uppdrag-till-verket-for-innovationssystem-att-inratta-ett-program-for-avancerad-life-science/.
[169]. Ibid.
[170]. Government Offices of Sweden, “Excellent research and innovation are encouraged in largest-ever research and innovation bill.”
[171]. “About us,” TestaCenter, accessed November 13, 2025, https://testacenter.com/about-testa-center/; “About us,” BioVentureHub, accessed November 13, 2025, https://www.azbioventurehub.com/about.html; “Innovation Hub,” NorthX Innovation, accessed November 13, 2025, https://www.nxbio.com/innovation-hub/.
[172]. Business Sweden, “INCENTIVES GUIDE: For companies looking to invest in Sweden” (Stockholm: Business, 2022), https://www.business-sweden.com/contentassets/41249a64a04e4309b7637b00c526eef8/incentives-guide.pdf.
[173]. “Health and life sciences,” Vinnova, accessed November 13, 2025, https://www.vinnova.se/en/innovation/life-sciences/.
[174]. Ibid.
[175]. “Life Sciences – the science of all living things,” Knut and Alice Wallenberg Foundation, accessed November 13, 2025, https://kaw.wallenberg.org/en/life-sciences-science-all-living-things.
[176]. Roschier, “Sweden modernizes its patent system,” news insights, November 26, 2024, https://www.roschier.com/newsroom/sweden-modernizes-its-patent-system.
[177]. “Intellectual property rights and intangible assets for Life Science,” Swedish Intellectual Property Office, last modified October 9, 2024, https://www.prv.se/en/students-and-researchers/doctoral-student-and-researcher/digital-courses-for-doctoral-students-and-researchers/intellectual-property-rights-and-intangible-assets-for-life-science/.
[178]. Erika Färnstrand Damsgaard and Marie C. Thursby, “University entrepreneurship and professor privilege,” Industrial and Corporate Change 22, no. 1 (2013): 183-218, DOI: 10.1093/icc/dts047.
[179]. Government Offices of Sweden, “Agreement with United States promotes cancer research,” press release, March 18, 2024, https://www.government.se/press-releases/2024/03/agreement-with-united-states-promotes-cancer-research/.
[180]. Government Offices of Sweden, “Uppdrag till Socialstyrelsen att samordna det nationella arbetet med genomförandeavtal med USA på cancerområdet” (Stockholm: Government Offices of Sweden, November 2024), https://www.regeringen.se/contentassets/6ec747a9d6b64fd39910fc5b24e8c4ae/uppdrag-till-socialstyrelsen-att-samordna-det-nationella-arbetet-med-genomforandeavtal-med-usa-pa-canceromradet.pdf.
[181]. Government Offices of Sweden, “Ändring av uppdraget till Socialstyrelsen att samordna det nationella arbetet med genomförandeavtal med USA på cancerområdet” (Stockholm: Government Offices of Sweden, June 2025), https://regeringen.se/contentassets/991bed74c831492ca46db5c0ba8eb9ac/andring-av-uppdraget-till-socialstyrelsen-att-samordna-det-nationella-arbetet-med-genomforandeavtal-med-usa-pa-canceromradet.pdf.
[182]. Association of the British Pharmaceutical Industry (ABPI), “Creating the Conditions for Investment and Growth,” (London: ABPI, December 10, 2025), https://www.abpi.org.uk/publications/creating-the-conditions-for-investment-and-growth/.
[183]. Alice Inch, “Life Sciences Sector Plan: what you need to know,” NHS Confederation, July 16, 2025, https://www.nhsconfed.org/publications/life-sciences-sector-plan.
[184]. Jacob Smith, “UK is Europe’s Leading Biotech Market, BIA Report Shows,” Drug Discovery World, January 2026, https://www.ddw-online.com/uk-is-europes-leading-biotech-market-bia-report-shows-40208-202601/.
[185]. BioIndustry Association, “UK biotech finishes on high despite challenging 2025, BIA report finds,” January 26, 2026, https://www.bioindustry.org/resource/uk-biotech-finishes-on-high-despite-challenging-2025-bia-report-finds.html.
[186]. Lucy Walters, “Top 6 Pharmaceutical Companies in the UK 2024,”PharmiWeb.Jobs, April 30, 2024, https://www.pharmiweb.jobs/article/top-6-pharmaceutical-companies-in-the-uk-2024.
[187]. Autolus Therapeutics, “Autolus – Developing CAR T Cell Therapies for Patients with Cancer and Autoimmune Diseases,” https://www.autolus.com/.
[188]. Ryan, “Patent Box,” https://ryan.com/europe/practice-areas/innovation-funding/patent-box/.
[189]. Fawz Kazzazi et al., “Evaluating the Impact of Brexit on the Pharmaceutical Industry,” Journal of Pharmaceutical Policy and Practice Vol. 10 (October 4, 2017), https://pmc.ncbi.nlm.nih.gov/articles/PMC5628446/.
[190]. Connor Lynch, “Part 2: What Impact Has Brexit Had on UK Drug Discovery?” Drug Discovery World, July 2023, https://www.ddw-online.com/part-2-what-impact-has-brexit-had-on-uk-drug-discovery-24962-202307/.
[191]. Kazzazi et al., “Evaluating the Impact of Brexit on the Pharmaceutical Industry.”
[192]. UK Department for Business and Trade, Department of Health and Social Care, Department for Science, Innovation and Technology, and Office for Life Sciences, “Life Sciences Sector Plan,” (London: UK Government, July 16, 2025), https://www.gov.uk/government/publications/life-sciences-sector-plan/life-sciences-sector-plan.
[193]. ABPI, “Creating the Conditions for Investment and Growth.”
[194]. UK Government, “Life Sciences Innovative Manufacturing Fund (LSIMF): Expression of Interest,” https://www.gov.uk/government/publications/life-sciences-innovative-manufacturing-fund-lsimf.
[195]. Zoey Becker, “UK lays out strategy to become a top 3 life sciences economy by 2035,” Fierce Pharma, June 23, 2025, https://www.fiercepharma.com/pharma/uk-lays-out-strategy-playbook-becoming-top-3-life-sciences-economy-2035.


