How E-labels Can Support Trade and Innovation in ICT, Medical, and Other Products
Displaying regulatory and other product information electronically is more practical than using small, confusing physical labels. But countries need to align their approaches to “e-labels” in order to maximize their benefits and avoid creating a new barrier to trade and innovation.
KEY TAKEAWAYS
Key Takeaways
Advantages of Using E-Labels 9
E-Labeling’s Use Around the World. 12
Recommendations for E-labeling Best Practices 15
Appendix: Country Case Studies 18
Introduction
Twenty-one countries—from China and the United States to Samoa and India—allow firms to use e-labels to show radio and telecommunication compliance marks for telecommunication products (e.g., smart phones). Several other countries allow them for medical devices to convey information and directions for product use. Countries may allow e-labels as part of emerging cybersecurity labeling schemes. The ease in which users can access product information means that e-labels can be used in many ways.
As more countries allow or consider allowing e-labels for regulatory purposes, there is the risk that divergent and complicated approaches will undermine their benefits and create a technical barrier to global trade in ICT and other products.
There are three main types of e-labels: a digital display on a device’s screen, a link to a website via a text URL, or a machine-readable code (e.g., a QR code). E-labels can contain a broad range of information beyond conformity-compliance labels. They can include a technical description of the equipment, such as instructions for proper use and service, warranty, equipment selection, and installation information.
E-labeling is not a way to avoid regulation. It is simply a way to convey information to regulators and consumers in a format that is much clearer and more easily understood (as compared to a confusing array of physical labels jammed onto a piece of a product). Governments, firms, and consumers all benefit from e-labeling. For example, regulatory and customs agencies can easily see conformity markings online, which firms can easily update in order to reflect regulatory changes. Consumers benefit, as they can easily see that the equipment is certified and suits their needs, and access other information about service requirements, upgrades, product warranty, and recycling. Equipment installers benefit, as they can access the most accurate and up-to-date technical data about the product and how to install it. Empowered by ubiquitous connectivity, all consumers and users expect to use technology to choose when, where, and how they engage with products they are investigating, buying, renting, using, and servicing.
The number of firms using e-labeling instead of physical labels for regulatory purposes is low, but growing. E-labeling is still relatively new, so its limited use is understandable. For example, the United States only started allowing it for telecommunication products in 2017. And several firms use both electronic and physical labeling as they transition to e-labels. However, more firms will inevitably use e-labels now that more countries allow them. Despite their low adoption, most if not all the major ICT producers use e-labels, which, in turn, covers most consumer electronics markets in Australia, China, Japan, Singapore, and the United States, among other countries. The use of e-labeling in the three main categories of consumer electronic goods (telephones, computers, and TV/radio/multimedia products) means it covers 60 percent of the consumer electronic market.[1] Indicative of this, one study estimates that in 2015–2017, e-labels were used in an estimated 78–90 percent of smartphones sold in Australia, Singapore, and the United States and 82–86 percent of computers sold in Australia and the United States.[2]
However, the growing use of e-labels also raises the prospect of countries enacting restrictive and conflicting requirements. If countries make e-labeling requirements overly complicated and prescriptive, and substantially different from other countries, they will undermine their primary benefit of greater simplicity and efficiency. There is a growing urgency for policymakers to ensure that their respective approaches align. Otherwise, firms face the daunting process of trying to abide by a proliferating range of e-labeling schemes, which may create a similar burden to the one they’re trying to avoid in the case of physical labels. It’d be a disappointing outcome if technological innovation were inhibited as firms had to alter product designs to make space for physical and e-labels.
The use of e-labeling has evolved since the Information Technology and Innovation Foundation’s (ITIF’s) last report, “How E-Labels Can Support Trade and Innovation in ICTs,” in 2017.[3] This report provides an updated analysis of how e-labeling can benefit regulators, consumers, and manufacturers alike. It examines the growing use of e-labels worldwide in more sectors, and the benefits of e-labels. Finally, the report presents a range of recommendations and best practices for how to enact an e-labeling system. Appendix A includes a comprehensive list of countries and a description of e-labeling policies, which includes an analysis of good and bad practices.
A summary of the recommendations for policymakers who are considering e-labeling:
▪ Governments should allow firms to voluntarily use e-labels to demonstrate regulatory compliance, such as via digital displays and QR codes.
▪ Governments should allow firms to use e-labels in as many products as possible. Smartphones and other devices with a screen are a logical place to start. However, with a growing number of digitally savvy consumers and regulators, countries should consider e-labels for a broader range of regulatory issues.
▪ Governments should develop a coordinated approach to e-labeling by ensuring all the various agencies (e.g., telecommunication regulators and customs authorities) are involved and working together.
▪ Countries should use an open and transparent rule-making process when developing their e-labeling frameworks to ensure their approach aligns with industry and global best practices.
▪ E-labeling is about streamlining and simplifying the delivery of information about regulatory compliance. Governments should set minimum requirements and be flexible in terms of how firms decide to display their e-labels.
▪ Countries should ensure that firms provide regulators, installers, and consumers with clear instructions about using e-labels.
▪ Countries should allow firms to host, control, and maintain the website with the regulatory and other information that e-labeling requires as they are best placed to manage this information (as opposed to a government-run database). Ideally, countries would work together to designate a single third-party to manage and audit the information.
▪ Countries should use international standards for e-labeling and associated technologies (e.g., QR codes), as they provide a common technical approach for firms to meet across multiple markets, and build interoperability between different regulatory frameworks. This supports innovation as firms only have to build to one—and not several—technical standard when designing products.
▪ Countries should use regional economic organizations to share best practices about e-labeling to support interoperable or harmonized approaches to e-labeling. For example, Southeast Asian nations should use Association of Southeast Asian Nations’ (ASEAN’s) Consultative Committee for Standards and Quality to develop a regional approach to e-labeling.
The Growing Benefits of E-Labels and Challenges to Further Adoption
E-labeling’s main use has been for compliance markings for a country’s telecommunications requirements, serving essentially two audiences: regulators and consumers. But it remains an open question as to how much attention consumers give to physical labels. As some electronic products get smaller, it is becoming a massive challenge for firms to include the current range of physical labels required across key markets, never mind adding new labels for environmental, cybersecurity, and other telecommunication reasons. For example, some countries (e.g., Chile and Colombia) are making firms add labels for smartphones to disclose information about the types of networks in which these devices operate (i.e., 2G, 3G, and 4G), their functionalities, and their compatibility with emergency alert systems.[4] Some countries also require these new, country-specific labels to be in the local language (which is problematic when a product is designed for many markets).
QR Codes: The Rise of a Ubiquitous Technology That Could Greatly Expand the Use of E-labels
People around the world have gained familiarity with machine-readable codes, especially QR codes, during COVID-19, which have become common due to the need for contactless interaction and contact tracing. For example, it became standard practice for restaurants to leave a QR code on tables so that customers could easily pull the menu up on their phones. The rise in consumer, business, and government awareness could be a powerful catalyst for e-labeling in more countries and sectors.
Policymakers previously cited a perceived lack of consumer and regulatory awareness of QR codes as a reason to avoid e-labeling, but widespread smartphone ownership and the massive growth in the use of QR codes during COVID-19 has clearly removed this as an issue in many countries.
A QR code is a type of matrix barcode (or two-dimensional barcode). In practice, QR codes often contain data for a locator, identifier, or tracker that points to a website or application. A QR code allows a scanning device or smartphone to direct the user to a website to view the labels, statements, and other relevant equipment information. Codes are printed on the equipment, viewable on the equipment’s screen, or affixed to its packaging.
Photo 1: Examples of hybrid traditional and e-labels with a QR code that links to a website with clear, detailed, and legible regulatory and other information[5]
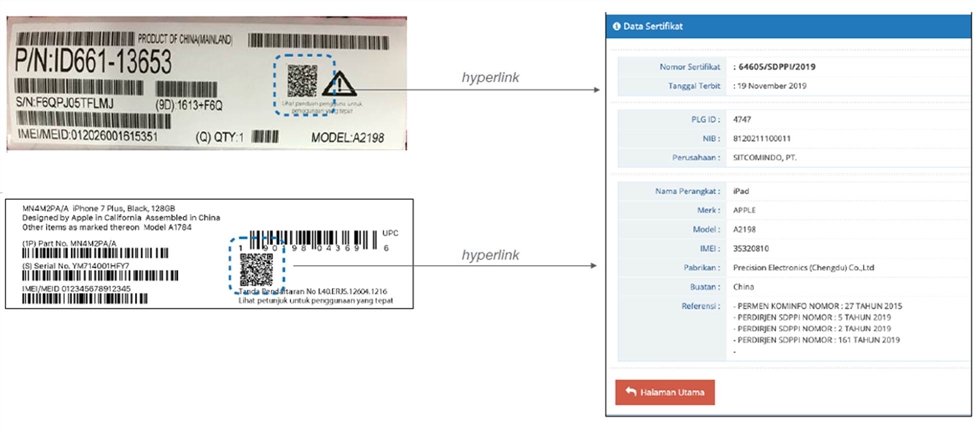
Governments previously cited a perceived lack of consumer and regulatory awareness of QR codes as a reason to avoid e-labeling. This, in part, was due to the fact that QR codes previously required users to download an app to scan them, which put people off from using them. This is no longer the case. In 2017, Apple updated its products to embed a QR code scanner into its iPhone’s camera app, and other smartphone manufacturers soon followed suit. With this change, QR codes became more accessible to many people around the world.[6] Measures related to COVID-19 also contributed to QR usage and familiarity skyrocketing to a 96 percent increase in reach and 94 percent increase in interactions from 2018 to 2020.[7] This trend is global: People in Australia, Canada, France, Hong Kong, India, Indonesia, Malaysia, Singapore, Switzerland, Thailand, and the United States widely use QR codes.[8] QR codes have become a recognizable, widely used, and accepted means of sharing information. According to a 2020 MobileIron poll of people in the United States, United Kingdom, and elsewhere in Europe, 83 percent of respondents had scanned a QR code at least once, and 72 percent scanned a QR code within the previous month.[9]
Firms are using QR codes in a variety of ways. Popular apps such as Snapchat, Spotify, and Venmo began implementing QR codes so people could quickly connect with other users, thereby furthering their general presence and proving their wide-ranging usefulness. The use of QR codes is vast, and already they are being utilized for electronic payments, coupons, marketing, education, and security, among many other applications. The widespread use of QR codes by consumers, businesses, and governments should address concerns about whether users would know how to access and use them.
Sectors Using E-labels
E-labels are being considered and used to address regulatory considerations in several industries.
Telecommunications Compliance
Government regulatory agencies most commonly use e-labels as a way for firms to show mandatory conformity marks for radiocommunications, telecommunications, electromagnetic compatibility, and electromagnetic radiation equipment standards. Hence, e-labels are most commonly used for smartphones, tablets, laptops, and other ICT devices. These labels convey information to consumers and keep companies honest about their products’ regulatory compliance.
Photo 2: Examples of physical and e-labels: (left) how an e-label appears on an iPhone, (top right) the labels on the back of a power adaptor for a laptop, and (bottom right) an example of physical labels for ICT products
Canada, the European Union (EU), the United States, and others allow firms to use e-labels for medical devices, mainly to provide users with a more convenient and helpful way to access Instructions/Directions for Use (IFU/DFU). For example, the EU allows e-labeling for implantable medical devices and their accessories, medical devices and their accessories with built-in screens, and stand-alone software (that is considered a medical device).[10]
Cybersecurity
Governments worldwide are considering adopting cybersecurity labeling as a way to communicate security features in ICT products and services to users in order to build understanding, confidence, and trust.
Using e-labeling to convey cybersecurity information is relatively new. Few countries have enacted cybersecurity labeling frameworks, and those few don’t allow e-labeling. For example, Singapore has a voluntary cybersecurity certification scheme and labeling proposal for Internet of Things (IoT) products. In 2021, Singapore made it mandatory to have certification/labeling for residential gateway devices, but firms could only use physical labels.
Governments are starting to consider cybersecurity labeling to convey security features in ICT products and services. While labels may help inform consumers about security features, they should not be perceived as a substitute for underlying processes that build security and trust.
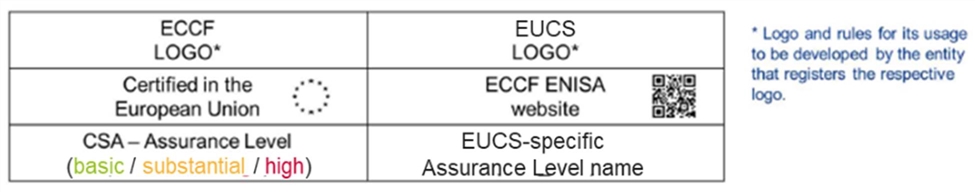
But e-labeling may be allowed elsewhere as other countries consider cybersecurity labeling. The United Kingdom’s consumer IoT code of conduct has a certification proposal, with the top three security requirements being mandatory. The United Kingdom is considering labeling requirements but has not yet announced details. The EU’s cyber-certification scheme for cloud (EUCS) includes a labeling requirement for firms to display for cloud services (photo 3). The draft label uses a QR code to access the European Union Agency for Cybersecurity (ENISA), which hosts certification information.[11] The Biden administration’s executive order on improving cybersecurity tasks the U.S. National Institute of Standards and Technology (NIST) and the Federal Trade Commission (FTC) to develop labels for consumers for IoT devices and software, which could potentially include e-labels.[12]
Photo 3: Demo label for the EU’s Cybersecurity Certification Framework[13]
It would be great if consumers considered device security as a part of their purchasing decision. Before making a purchase, consumers often have limited insight into the security features of a finished product, device, or service. Providing users with clear information about firms’ adherence to cybersecurity standards and specific topics (e.g., the security features and functionality in devices or services) can foster market competition (based on security), build trust, and help them follow good cybersecurity practices. Putting more information about product security in e-labels will make it easier for online retailers to provide this information to consumers when they shop online and compare products.
Policymakers need to be careful in using labels for cybersecurity as it is far more dynamic than other regulatory requirements. While a cybersecurity-specific label encourages firms to adopt the underlying security features, practices, or certifications, policymakers should not see the label as a substitute for the time and effort firms dedicate to building security and trust into a product.[14] Furthermore, as with Singapore’s “four-star” ranking system, countries should be careful not to mislead consumers with systems that consumers (may) think mean a product with the highest ranking is perfectly secure.[15] The dynamic nature of cyberthreats and cybersecurity means that a static ranking on a physical label may soon be out of date, whereas an e-label that seeks to inform users about a firm’s ongoing efforts to secure products provides a more realistic understanding of a product’s cybersecurity status. In this way, the dynamic nature of e-labels becomes apparent, as firms can quickly and constantly update them.
Advantages of Using E-Labels
The benefits of using e-labeling have become more apparent since ITIF’s last report in 2017, especially given how consumers and regulators have become more digitally savvy. When designing e-labeling policies, policymakers should keep these benefits in mind, as they apply the principles and best practices discussed in the final section of this report.
Greater Information and Utility
Products overcrowded with physical labels confuse end users (see photo 1). Manufacturers, regulators, customs officials, and consumers are left to make sense of myriad conformity assessment and certification marks and other labels crammed onto a product.
E-labeling enables users (government regulators, consumers, and commercial users) to get the information they need in a way they want it (rather than after purchase and unpacking). This comes as firms need to provide more and more information to end users as equipment becomes more complex. Accessibility via electronic labeling removes the physical space constraint of products that get smaller and smaller, which makes it difficult to decipher what the labels are and what they mean (see photo 2). It is also in a more readily understandable format—with clear and detailed text, graphics, and videos on a phone, tablet, or computer screen.
E-labeling makes it much easier for firms to provide more information, versus the limited amount of information conveyed by physical labels on products, associated packaging, and document inserts. For example, it allows firms to provide detailed installation information. It also makes it much easier for firms to provide this information in more languages. Similarly, the end user can assess whether the equipment meets their performance, contract, and installation requirements.
A significant advantage of e-labeling is that it enables users—whether government regulators or consumers—to get more of the information they may need and provide it when they want it and on the device of their choice.
Consumer and User Friendly
E-labels empower consumers with more readily understandable information. Finding the right instructions in lengthy multilingual booklets can be challenging given the small fonts used in user manuals. E-labels also make it easier for users to learn what after-sales service and maintenance the product may need and programs/services that may assist in providing them. E-labels also allow firms to provide a detailed explanation of warranty terms that otherwise may be incompletely summarized on physical product labels or discarded as part of user manuals provided (and soon lost) after purchase. E-labeling also allows firms to quickly respond to increasingly rapid changes of technical data that determine equipment suitability and selection, avoiding the scenario wherein printed labels can soon go out of date. In other words, e-labels can help ensure that the relevant information provided to consumers is the most accurate and up to date possible (and can handle versioning history in case of any changes). For example, the dynamism of e-labels is particularly well suited to the constantly changing nature of cyberthreats and security, thus allowing consumers to see how firms are continually working to ensure their products are safe and secure.
While a product may be small, it may require complex and extensive instructions. Requiring these in paper booklets for small products is becoming a growing cost. E-labels allow firms to direct users to websites to access these instructions, demonstration videos, and interactive FAQs. Electronic instructions also lend themselves to better instructions, given they allow firms to use animations and videos.
Smoother and Quicker Product Delivery
Using physical labels slows down and complicates product deliveries whenever firms need to affix country-specific physical labels, especially if there are sudden changes in supply and demand. For example, no one in Brazil wants a phone with Chile’s labels pasted on it. Firms are likely to be more cautious in supplying markets that require unique physical labels and not over-commit products for those markets, given the cost and complications of affixing physical labels. This means consumers in markets with burdensome labeling requirements are likely to face delays in getting the latest products given the extra work firms need to do to get their products ready for that market. Ultimately, consumers may go elsewhere to buy products, such as through black/gray markets or other countries.
E-labels Allow for Easier Enforcement
Regulators and customs officials can easily access compliance information via e-labels, which has the benefit of helping firms avoid customs-related shipment and delivery delays due to issues with certification compliance. A master list of labels and compliance information on the Internet or the device, kept up to date by manufacturers, offers real-time compliance information far beyond a simple mark on a tiny label.
Reduced Environmental Impact
E-labels provide several environmental benefits. E-labelling supports broader work around the “circular economy” and other environmental initiatives (e.g., more efficient industrialization processes).[16]
E-labels allow manufacturers to reduce the material they use in labels, especially with conformity certification changes and product recalls when firms need to replace and update them. If a firm conveys inaccurate information, or if the technical data for equipment or a given system change, firms can quickly correct and update relevant e-label information, rather than recalling products and removing and replacing physical labels. Firms can also provide customers with detailed information about how they can physically dispose or ensure proper recycling of their products.
E-labels support good environmental outcomes. Replacing physical labels for millions of devices is a huge waste; e-labels can provide advice about how to properly dispose of or recycle equipment; and replacing paper instructions with virtual ones saves a lot of paper.
E-labels also save firms considerable costs, as they make it possible to provide virtual instructions sheets. Paper-based product labels and manuals are resource-intensive—both from a manufacturing and an environmental perspective. Having to print materials in multiple languages compounds the problem. For example, the EU’s Medical Device Regulation requires user manuals to be printed in all 24 European languages—which takes time and thus has a detrimental impact on speed to market.
Reduced Impact on Product Innovation and Product Costs
Technological innovation means ICT, medical, and other products are shrinking in size and becoming more complex and capable, such that physical labeling requirements become a constraint on product design. Manufacturers may need to alter the optimal product design just to satisfy labeling requirements.
E-labeling supports trade and innovation of ICT and other products as it’s easier and cheaper for firms to use. Manufacturers spend significant amounts of time and money on creating, controlling, maintaining, and producing product markings, packaging, and instruction sheets used to convey certification marks and other product information. These costs increase when manufacturers modify labels, rework products, and perform in-country retrofits due to changing labeling requirements.
Firms want to build only one (or as few as possible) version to satisfy as many different conformity requirements as possible. E-labels help firms save money in the event of changes in conformity assessment requirements or product recalls, given the cost of replacing physical labels and redesigning products. E-labeling also reduces time to market for new products, as engineers don’t need to speed time figuring out the increasingly complicated task of physically showing a range of different compliance marks. Firms also save considerable money as e-labels can lead to fewer calls to technical and customer service departments by allowing users to access and understand a wide range of product information.
The cost savings from moving from physical to e-labels are significant. For instance, according to one estimate, the introduction of e-labeling for radiofrequency devices (i.e. smart phones) in the United States allows manufacturers to save over $80 million a year.[17] Similarly, a study by VVA (an economic consultancy firm) estimates that the cost of indicating compliance for the consumer electronics industry in Europe are around $929 million per year, and allowing e-labeling would reduce these costs by approximately 15 percent ($139 million).[18]
Common Concerns Policymakers Cite in Considering E-labeling
Policymakers cite two common concerns in considering allowing e-labeling: regulatory reluctance to change and getting government agencies to cooperate in allowing e-labeling. As already discussed (and rebutted), a previous concern voiced by governments about e-labeling was that consumers might not understand and thus won’t use e-labels. But consumers around the world have shown they are tech-savvy.
Regulatory Reluctance to Change
In many countries, telecommunication regulators and customs agencies are reluctant to abandon their traditional reliance on inspecting physical labels for import inspections and post-market surveillance. It represents a clear and tangible way to know that the products meet local requirements. This is understandable. In the 20th century, applying a physical label to products was an obvious solution to ensure they met local requirements. However, the growth in global production networks, the shrinking size of ICT products, and the option of using digital technologies mean physical labels are no longer the best approach. Burueactic reluctance stems from the fact that e-labeling disrupts the status quo and forces agencies to change regulations, inspection procedures, and other processes.
Regulatory reluctance to change from physical to electronic labels is the main barrier to adoption. Regulators (misguidedly) think e-labels undermine their authority, yet the fact that this hasn’t happened in the dozens of countries that use e-labels shows that this is not true.
Regulators are often reluctant to change, as they think it will undermine their supervisory authority. However, the growing use of e-labels in countries and sectors worldwide shows that this is not true. For example, according to the Australian telecommunications regulator, e-labels have not affected their market surveillance. The presence of a label on a device does not mean that device is compliant; it is the compliance documentation (test reports, Declarations of Conformity, certification body statements, etc.) that demonstrates compliance.[19] And e-labels make it easy for regulators to verify these marks and documents.
Similarly, e-labeling does not necessarily mean governments need to spend more on compliance. For example, the U.S. Congressional Budget Office estimated that implementing the E-LABEL Act would have a negligible effect on net discretionary costs at the Federal Communications Commission (FCC).[20]
The challenge for firms and policymakers that support e-labeling is working with reluctant regulators to facilitate policy reforms. Hence, it’s helpful to layout a detailed and suitably long-term plan of action that brings regulators and industry together on the design, testing (e.g., through pilot programs), implementation, and review of changes. Such a step-by-step approach will help overcome regulators’ reluctance to change.
Getting Government Regulators and Agencies to Cooperate
Countries may be reluctant to enact an e-labeling framework, as it involves cooperation and buy-in from multiple government ministries and agencies. For example, a telecommunication regulatory agency or a medical/health regulator may need to work with its respective ministry and the customs agency to get their approval and cooperation to allow e-labeling. Navigating multiple bureaucracies means there are numerous opportunities for officials who may not support e-labeling to obstruct reforms.
E-Labeling’s Use Around the World
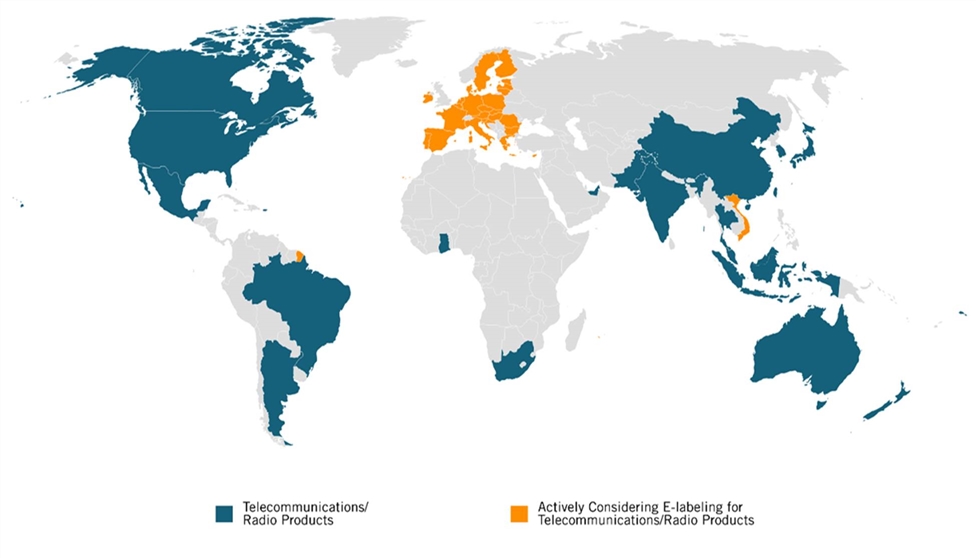
Most of the world’s major economies allow firms to use e-labels for regulatory purposes (see figure 1 and the appendix for a detailed list). As of 2021, 21 countries allow firms to use e-labels to show radio and telecommunication compliance marks, while several other countries allow medical device manufacturers to use e-labels to convey information about how to properly use devices.
Figure 1: Map of countries that allow, or are considering, e-labels to display telecommunication/radio compliance marks
E-labeling in the Asia Pacific
Southeast Asia is home to many consumers and production centers that use e-labeling, yet ASEAN has thus far not added the issue to its agenda. Half of ASEAN’s members (Malaysia, the Philippines, Singapore, Thailand, and Vietnam) allow or are considering e-labeling. ASEAN and its member countries have long worked on regional standards and conformity assessment-related issues, mainly via ASEAN’s Consultative Committee for Standards and Quality (ACCSQ). There’s the potential for ACCSQ to add e-labeling to its agenda. For example, in 2019, ASEAN created a digital trade working group under ACCSQ based on recommendations from the ASEAN-AustraliaDigital Trade Standards Cooperation Initiative.[21] However, thus far, e-labeling has not been part of early discussions.[22]
Southeast Asia is home to many countries and consumers that use e-labels, yet the region is not yet considering a regional approach that would support ICT trade and innovation and good regulatory outcomes.
APEC’s bigger and broader membership includes many members that allow e-labels (China, Japan, Singapore, and the United States, among others). Yet, e-labeling has not been on the APEC agenda. APEC’s Sub-Committee on Standards and Conformance (SCSC) covers many similar issues as ACCSQ in aligning standards and conformity assessment processes. The 2017 ITIF-Commerce Department workshop on e-labeling took place alongside an SCSC meeting. APEC’s Telecommunications Working Group and the Digital Economy Steering Group also work on issues related to e-labels. SCSC should aim to work with these groups to share best practices and facilitate regional alignment around e-labeling.
International Standards for E-labeling
With international standards, technical and regulations experts work together in an open, consensus-based process that lends itself to developing the best approach to address particular technical issues. International standards are designed to support domestic laws and regulations in many countries. International standards provide regulators and firms with tools to address common requirements related to a product or regulatory issue. In most countries, adopting international standards is voluntary; however, regulators often recognize the use of international standards to meet technical regulations, and conformity assessment bodies will review them and associated technical files and data while assessing a firm’s/product’s compliance.
For example, there is already an international standard for QR codes, as it’d obviously be hugely problematic if different countries and firms used different types of QR codes. ISO/IEC 18004:2015 defines the requirements for the symbology known as QR Code. For medical devices, the International Medical Device Regulators Forum (IMDRF), which is made up of health regulators from the world’s major countries, issued guidance on the principles of labeling for medical devices, which defines e-labels and when and how they can be used.[23] This guidance represents consensus of labeling best practices from a regulatory perspective. It is not binding, but adherence to it can make auditing and compliance easier for firms. An international standard for e-labels is near completion—awaiting final edits and approval—and should be finalized and published before the end of 2021. Industry, government, and other experts from around the world have been working to develop it at ISO and IEC forums.
The standard involves two core standards:
▪ ISO/IEC 22603-1, Information technology—Digital representation of product information—Part 1: General.
▪ ISO/IEC 22603-2, Information Technology—Digital representation of product information—Part 2: Requirements for electronic devices with integral display.
Recommendations for E-labeling Best Practices
E-labels are a valuable tool for governments, firms, and consumers. However, in allowing e-labels, governments must follow emerging best practices to ensure they maximize the potential benefits.
Allow Firms to Use E-labels, Such as QR codes, as an Option to Show Regulatory Compliance
Governments should allow firms to voluntarily (not mandatorily) use e-labels to demonstrate regulatory compliance. For example, telecommunications regulators should allow firms to use e-labels as an alternative to physical markings on ICT products.
QR codes should be part of the discussion as countries look to allow e-labels or expand existing systems. While e-labels should be technology neutral (given we don’t know whether there’ll be a new technology to supersede QR codes, for example), countries should specify that QR codes are one way for firms to show e-labels. As discussed, QR codes offer a relatively quick and easy way to connect a regulator or consumer to a website with compliance and other information.
Specify Which Products Can Use E-labeling—but Consider Using E-labels Broadly
Countries should consider using e-labeling broadly given ubiquitous smartphone ownership and how familiar consumers are with QR codes and the Internet. Most countries using e-labels have started by allowing it on devices with built-in screens, which is understandable given the nature of such products. Policymakers could start there and build on it with other products.
E-labeling should be an iterative process that regulators build on over time as firms and consumers become comfortable with e-labels. Whether a product has a screen is less important now, given how widespread smartphone and computer ownership is. Furthermore, consumers are increasingly reliant on accessing product information and manuals over the Internet, partly as they prefer this over bulky manuals with small fonts they typically discard immediately after purchase.
Ensure Clear Leadership and Coordination Between Agencies When Developing an E-labeling Frameworks
Governments that decide to allow e-labels should ensure that they designate a lead agency and make it clear that related government ministries and agencies must work with them in developing and enacting an e-labeling framework. It can be difficult for a specific regulator, such as a telecommunications regulator, to enact e-labeling if their respective ministry and customs and border officials are not supportive or engaged in the process. Coordination is essential when multiple agencies in a country consider allowing e-labeling in their respective areas. This issue depends on each country, as institutional arrangements differ by country. For example, the agency responsible for electromagnetic and radio transmitting devices may be separate from the agency involved in safety, environmental, and other regulatory compliance issues. It’s nonsensical for countries to have multiple different and potentially conflicting e-labeling requirements.
Run an Open and Transparent E-labeling Development Process
Countries should set up a transparent rule-making process when developing their e-labeling framework to ensure it aligns with global best practices. The process should be open to public and industry comments and engagement, including several opportunities for feedback and engagement. An open and participatory process is essential, as a seemingly small mandatory requirement may have a significant cost and design impact on firms. Governments also need to provide a reasonable transition period, as firms need time to assess regulatory changes and apply them to their design and production processes for new products.
Set Minimum Requirements, but Be Flexible
For example, e-labeling minimum requirements could include the basic details already commonly found with current products, such as conformity labels and statements; product details (including manufacturer details and contact information); and the product name, model, features, and production date. Just as multiple physical labels act as a technical barrier to trade, so too could multiple prescriptive and unique e-labeling requirements. Furthermore, a flexible e-labeling regime allows a country to adapt quickly to innovations in labeling and to new technology that might not easily accommodate a particular type of label in the future.
Ensure E-labels Use Clear and Common Terminology and Information Requirements
As part of setting minimum requirements related to what information they want firms to show via e-labels, countries should use clear and common terminology to ensure that definitions and other information details are consistent with international best practices. For example, if a country requires a firm to show the country of manufacturing, then specify what this means and ensure that it reflects the firm that actually makes the product. Some countries require firms to specify where a product is designed, assembled, and imported. Where possible, countries should use relevant international standards that specify what e-documentation firms should provide. For example, ISO 20417:2021 sets out what information medical device manufacturers need to provide.[24]
Ensure Firms Support E-labels for a Reasonable Period of Time
Countries should ensure that firms provide information via an e-label for the lifecycle of the equipment, including for a reasonable period of time after the equipment has been permanently discontinued by the manufacturer. This may vary depending on the equipment, given some equipment (e.g., core telecommunication network equipment) may be used for decades.
Ensure Firms Provide Clear Information About How to Access Information
Countries should ensure that firms provide regulators, installers, and consumers with clear instructions about using an e-label to access regulatory compliance, installation instructions, maintenance, and other information. These instructions should accompany the equipment by being affixed to the equipment or on the equipment packaging or packaging material, or provided with the equipment documentation.
Provide Clear Guidance About Reporting and Hosting of E-label Information
Countries should allow firms (as opposed to a government agency) to host and maintain e-labeling information, as they’re best placed to manage and update this information.
Countries should set minimum requirements for hosting. Firms should be responsible for ensuring a working link between the e-label and the service hosting the relevant compliance information (e.g., a website). Countries should ensure that accessing e-label information does not require any fees or special access codes, the e-label information does not have any unnecessary copyright restrictions, and it can be accessed on a web browser.
Use International Standards for E-labels, QR Codes, and Other Associated Technologies and Requirements
For Regional Organizations, Explore Regional Approaches to E-labeling
Countries should use regional economic organizations to share information and best practices about e-labeling in order to build compatible (and not conflicting) approaches to e-labeling.
ASEAN and APEC should explore how they can help share best practices about e-labeling to encourage more countries to use them and to do so in a way that aligns with emerging international standards and best practices.
Southeast Asian nations should discuss e-labeling policies in ASEAN’s Consultative Committee for Standards and Quality working groups on standards, technical regulations, and conformity assessments. Indicative of the potential to work on e-labels, ACCSQ has successfully harmonized various standards related to automotive, electrical and electronic equipment, and medical devices. Similarly, APEC’s SCSC could host further workshops on e-labeling.
Conclusion
E-labeling is a modern tool for modern regulations and trade. It reflects how familiar regulators, consumers, and other users are with modern technology, and how comfortable they are accessing information digitally. However, the full benefits of e-labeling will not be realized until the majority of countries allow them in place of physical labels and more countries align their respective approaches to e-labels. Encouraging more countries to use e-labels for more issues will take time, effort, and careful consideration to ensure it is done well and does not inadvertently create a new technical barrier to trade and innovation akin to the one firms are trying to avoid with small, crowded panels of physical labels.
This report was made possible in part by generous support from the International Trade Administration (ITA) at the U.S. Department of Commerce. ITIF prepared this report with Thomas & Reed, LLC for ITA under U.S. Government Contract No.1331L521F13500386 to advance ITA’s work with the Association of Southeast Asian Nations Secretariat’s Consultative Committee on Standards and Quality and ASEAN Member States. The findings and conclusions in this document are those of the author, who is responsible for its content, and do not necessarily represent the views of ITA. No statement in this report should be construed as an official position of ITA or the U.S. Department of Commerce. ITIF retains the copyright to this report. The U.S. Government retains the unlimited right to use, disclose, reproduce, prepare derivative works for; distribute to the public copies of; and perform and display publicly for this report—in any manner and for any purpose, and to have or permit others to do so,.
The author thanks Rob Atkinson, Stephen Ezell, Daniel Castro, Malachy McLaughlin, and the government officials, private sector representatives, trade associations, and others who helped him understand the changing use of e-labeling around the world. Any errors or omissions are the author’s responsibility alone.
About the Author
Nigel Cory (@NigelCory) is an associate director covering trade policy at ITIF. He focuses on cross-border data flows, data governance, and intellectual property and how they relate to digital trade and the broader digital economy.
About ITIF
The Information Technology and Innovation Foundation (ITIF) is an independent, nonprofit, nonpartisan research and educational institute focusing on the intersection of technological innovation and public policy. Recognized by its peers in the think tank community as the global center of excellence for science and technology policy, ITIF’s mission is to formulate and promote policy solutions that accelerate innovation and boost productivity to spur growth, opportunity, and progress.
For more information, visit itif.org.
Appendix: Country Case Studies
The country case studies describe how key countries use (or, in some cases, don’t use) e-labels. It is a comprehensive list of countries that allow e-labels for telecommunication products. However, its coverage of countries that use e-labels for other uses is indicative (not comprehensive). These other use cases are included to highlight how different countries are using e-labels, such as QR codes, for such various regulatory issues as product safety and medical device instructions.
Argentina
Argentina has allowed e-labeling for ICT telecommunication products since 2015.[25] In 2021, Argentina’s telecommunication regulator (Ente Nacional de Comunicaciones) released updated labeling requirements for telecommunication that reinforced its existing rules allowing e-labeling.[26] However, in 2018, Argentina enacted a new regulation that required safety markings (S-type marking) on a range of ICT devices such as chargers, notebooks, routers, and monitors.[27]
Australia
Australia has allowed e-labels since 2010. In 2020, Australia enacted the Radiocommunications Legislation Amendment (Reform and Modernisation) Bill 2020, which allows QR codes, or a similar mechanism, if the relevant link is to information on a website.[28]
In 2016, roughly 80 percent of smartphones and PCs sold in Australia utilized e-labeling, and according to the Australian regulator, e-labeling has not affected the market surveillance process. The electronic label is considered an equally legitimate way to show regulatory information.[29] It is estimated that approximately 81 percent of smartphones and 78 percent of tablets sold in Australia in 2016 used e-labels.[30]
The introduction of e-labeling in Australia was mainly industry driven. Australian regulators have consistently used a participatory approach, involving all relevant stakeholders and several rounds of consultations for feedback and engagement, to design a “win-win” solution for the industry and other stakeholders impacted by the measure (i.e., market surveillance authority, customs agencies, and consumers).[31]
Brazil
Brazil adopted a flexible e-labeling regulation that is in line with international best practices. On April 22, 2020, ANATEL (Brazil’s Regulatory Agency for approving the certification for ICT equipment in Brazil) issued ACT 2221, updating the regulations for the marking of certified telecommunication products, which allowed for the use of e-labeling as one option.[32] ANATEL also allows the use of QR codes and the use of ANATEL’s physical label. It is only allowed for a product with a screen or that connects to an external monitor. The product must enable access to the e-label without codes, passwords, specific authorization, plug-ins, or special accessories. The e-label must be easily accessed by a user and take a maximum of three steps from the device’s main menu.
The instructions to access the e-label must be in the user manual, the quick guide pamphlet, product packaging, or the company website. The e-label must come already installed from manufacturing, be electronically protected, and remain recorded in the device’s memory even when the operating system is restarted or reset to the original manufacturing setting. When the e-label is for a device to be used directly by a final consumer, ANATEL’s certification must also be provided on the product packaging.
To use the QR code, firms must provide the pre-defined minimum list of information. The QR code must be accessible by any device with reading capacity, without the need to use any proprietary software. Again, ANATEL’s certification must also be provided on the product packaging. ANATEL also allows flexibility for scenarios when it is not possible to put the certification on the product due to space restrictions or technical difficulties, in which case the firm can apply for an exemption.
Canada
Canada began allowing e-labels in 2014. To use e-labels, a device must have a built-in screen, information must be provided on how to access the e-label, and it must be accessible in three or fewer steps. Devices without built-in screens can use an audio message or connected host device screen.[33]
On top of this, the United States-Mexico-Canada free trade agreement (USMCA) specifies that parties shall allow e-labeling to demonstrate electromagnetic compatibility and radiofrequency compatibility. Sector Annex 12-C specifies that “electronic labeling means the electronic display of information, including required compliance information.”[34]
Chile
Chile’s use of conformity markings is indicative of what not to do.
The March 2017 Resolution 1463 contains the labeling requirement. First, Chile’s approach to conformity markings for telecommunication products, such as smartphones, is problematic, as it only allows physical labels on product boxes and requires them to be displayed in stores and on e-commerce sites. Chile’s labels are particularly problematic because they are big and unique to Chile.[35] Chile requires a sticker on each imported phone to state the bands (3G, 4G, 5G, etc.) it is compatible with, as well as whether the device has Chile’s Emergency System Alert (see figure 2) installed. This is duplicative given all this information is in the product packages, manuals, and websites. The sticker itself has defined characteristics, and its placement is regulated. This requirement does not provide an additional benefit or improved information to consumers yet poses a high cost for firms to comply. Chile also enacted this regulation and design for the labels without any public consultation. Second, in 2018, Chile allowed firms to use QR codes to show electrical safety information in adapters for mobile phones. In 2020, it also allowed this for other electronics. However, the process to obtain the certification to use the QR undermines the benefits of using QR codes because it is so burdensome.[36]
Figure 2: Examples of the large and unique labels Chile requires[37]
China
E-labeling is widely used (voluntarily, driven by firms) in many retail stores. Chinese consumers are familiar with using e-labels, especially QR codes. From a regulatory perspective, China has allowed e-labels for devices with an unremovable, integral screen since 2015. Firms must provide users with instructions on how to access the e-label. The China Compulsory Certificate must still be provided on individual packaging of a litany of products.[38]
In 2019, China’s State Radio Regulatory Commission enacted rules for when a device has been registered at the Ministry of Industry and Information Technology (MIIT), a registration number and a QR code are issued, and must be made visible in the distribution store or on the website (if the product is sold online). If there is no store or website, the vendor shall keep the registration number and QR code. The customers may scan the QR code to obtain information about a product and its SRRC certification.[39]
The European Union
The European Union (207/2012) allows firms to use e-labels for communicating electronic instructions for the use of medical devices.[40] However, the EU is the last major economy to not allow e-labeling for telecommunication products. The EU’s product compliance and market access regime is governed by the New Legislative Framework, a package of legislation with various health and safety requirements. This includes the CE (the European conformity mark) marking obligation, which must be affixed visibly, legibly, and indelibly to products. The Blue Guide, which gives more practical guidance to the legislation, repeats this and states that electronic labeling alone is not allowed. Electronic waste regulation has a similar obligation for product marking with a “wheelie bin” icon.
Part of the problem is market surveillance authorities in EU member states also voice traditional (and misguided) concerns about e-labeling, such as it would be too complicated for users to switch on a device to check (as opposed to seeing a physical label) and Internet connectivity is not 100 percent. Indicative of this reluctance to change, only 42 percent of EU market surveillance authorities in a Digital Europe survey said they believe the e-labeling would make it easier to demonstrate compliance.[41]
European industry continues to make a case for the EU to join most of the rest of the world in allowing e-labels for regulatory compliance. In that same Digital Europe study, 76 percent of European businesses would adopt e-labels if it were available, and half of market surveillance authorities believe e-labeling would reduce their costs.[42] In a separate study, Valdani Vicari & Associati estimated that allowing e-labels in the EU would reduce the costs of indicating compliance for the consumer electronics industry by 15 percent.[43]
European industry supported an amendment that would’ve allowed e-labeling as part of the proposed Market Surveillance Regulation and as part of revisions to the Blue Guide. As to the former, the amendment was not approved. As to the latter, the European Commission said this was not possible and that the New Legislative Framework would need to be changed first. Attaching e-labeling reforms to such a large, complicated legislative framework makes it incredibly challenging to make progress in allowing firms to use e-labels for regulatory compliance in the EU.
Ghana
Ghana has allowed the use of e-labels in place of physical markings since 2015. It is the manufacturers’ responsibility that labels are tamper-proof.[44]
India
Devices with an integrated display screen may use an e-label so long as it is easily accessible and cannot be changed by a third party.[45] Devices using e-labels shall have a physical label on the packaging of the product at the time of import, storage for sale, and sale or distribution. For devices in bulk or not packaged individually, a removable adhesive label on the packaging is acceptable. Devices shall not require any special accessory/tool or additional plug-in (e.g., installing a SIM card) to access the e-label. The compliance-related information shall be programmed by the responsible party and the information shall be secured in such a manner that third-parties cannot modify it. The information can be in the firmware or software menu provided it is easily accessible and cannot be modified. The information shall be accessible in no more than four steps in a device’s menu. The “Standard Mark” displayed electronically shall meet all the relevant requirements of the guidelines issued by Bureau of Indian Standards circular (CMD 3/8:1/6975 of December 3, 2015).
Indonesia
In 2019, Indonesia’s Telecommunication Authority enacted new regulations that allow firms to use e-labels (e.g., a QR code) to show their regulatory mark.[46]
Japan
Japan has allowed e-labels since 2010 for devices with built-in screens. Firms must provider users with information about how to access the e-label.[47] In 2019, Japan’s Pharmaceuticals and Medical Devices Act was amended to allow for e-labeling in the health care sector.[48] A code may be printed on the outside of a device’s packaging to access the labeling information.
Malaysia
E-labelling has been allowed for devices with built-in screens since 2015, and “may be attached to the medical device itself, on its packaging or as a packaging insert or may be made available by other means, for example by electronic means.”[49] It is not allowed for external displays that connect to a product. Firms that use e-labeling must explain in accompanying documentation how the e-label can be viewed and retrieved.[50]
Mexico
Mexico’s national-level standard 16 provides the overarching general framework for e-labeling. However, it is up to each government agency (through their specific standards and regulations) to decide what information can be shown via physical or electronic labels. Since 2015, Mexico’s telecommunications regulator has allowed e-labels for ICT products with an integrated or tethered screen (via regulation IFT-008-2015).[51] Mexico is considering a new regulation that would allow some electrical safety information to be shown electronically, but this is still under consideration.
On top of this, the USMCA specifies that parties shall allow e-labeling to demonstrate electromagnetic compatibility and radio-frequency compatibility. Sector Annex 12-C specifies that “electronic labeling means the electronic display of information, including required compliance information.”[52]
New Zealand
E-labeling is permitted as long as accompanying documentation explains how to access the label, and it is applied in a way that is difficult to delete, modify, or prevent its displaying.[53] For medical devices, the instructions for use may be provided electronically through files supplied with the device on read-only media, files downloadable from the Internet, and help files displayed by the device—but upon request, a copy of physical instructions must be provided.[54]
Nigeria
Nigeria does not allow e-labels for telecommunication products.
However, in 2018, the Standards Organisation of Nigeria (SON) implemented new regulations relating to the Product Authentication Mark (PAM), requiring all imported products arriving at Nigerian ports to have met the requirements of the relevant Nigeria Industrial Standards or other approved specifications in Nigeria, meaning they must pass authenticity and traceability certification and display an appropriate PAM sticker. (See photo 4). Importers must ensure that the application for PAM Stickers has been submitted before final clearance from SON of the goods at the port of entry.[55]
Photo 4: Nigeria’s use of QR codes as part of its Product Authentication Mark[56]

Pakistan
Philippines
Although the Philippines does not have an explicit e-labeling law or regulation, the National Telecommunications Commission has reportedly allowed firms to use e-labels on a case-by-case basis.
Russia
Russia does not allow e-labels for telecommunication products.
However, in July 2019, Russia introduced a new law mandating the labelling of consumer goods that will involve e-labels, including digital RFIDs and QR codes. It will be phased in over time, and by 2024, all categories of consumer goods must be labelled and subject to traceability requirements. According to the law, consumer goods sold in Russia must be labelled with a special digital code that guarantees their authenticity. A dedicated digital track-and-trace system, called Chestny ZNAK, will manage the new digital labelling scheme. All consumer goods manufactured in Russia or destined for export to Russia must be labelled with a code that serves as a “digital passport,” tracking the product on its journey from factory to store.[57]
Singapore
Singapore has been relatively successful in using e-labels. The e-labeling process in Singapore has been government-led, with the regulatory agency setting the agenda and requirements and providing industry the possibility to comment.[58] Considering ICT products comprise almost one-quarter of the value of Singapore’s gross domestic product (GDP) (with telecommunications being the third-largest sector), the adoption of e-labels fits well with the country’s general regulatory approach, directed toward facilitating—and modernizing—trade.[59]
Singapore has allowed e-labeling for products with an integrated screen since 2012. Over 80 percent of the smartphones sold in Singapore from 2013 to 2016 utilized e-labeling.[60] Manufacturers need to provide an instruction guide or leaflet in the packaging accompanying the registered equipment describing the method it has adopted to display the compliance label and provide clear instructions on how consumers may locate the electronic compliance label for their own verification.[61] The Singaporean regulator also offers consumers and enforcement officers the ability to verify online whether the telecommunication equipment is registered through the “Equipment Search” function available at its Telecoms Licensing portal.[62] Singapore also allows medical devices that previously required thick leaflets of regulatory information to display their labels electronically.[63]
The Gulf Cooperative Council
In 2019, the Standardization Organization (GSO) of the Gulf Cooperation Council (Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, and the United Arab Emirates) developed and launched a new version of the Gulf Conformity Tracking System (GCTS), which allows QR codes to link to a registered product page online. This can be used for a broad range of household products, such as fans, fridges, food mixers, toasters, microwave ovens, and air conditioners.[64]
Samoa
Samoa allows firms to use e-labels for regulatory identification, but it requires the ID to be in the product manual and on the product box.
South Africa
Since 2013, e-labeling has been allowed on devices with an integral screen. The label must be accessible during the equipment’s power-up sequence, under the equipment’s system information page, or under the help menu.[65] However, e-labeling also must be requested in written form. The Independent Communications Authority of South Africa issues formal letters of approval.[66]
South Korea
Since 2015, South Korea has allowed e-labeling for devices with an inbuilt screen.[67] The 2016 Electrical Appliances Consumer Products Safety Control Act specifies that cell phones, smartphones, tablets, and notebook computers (as of July 2021) be included in this category. The manufacturer must indicate that their product uses e-labeling and provide instructions on how to access the label in less than three steps.[68] E-labels must include local suitability evaluation labels and identification codes for certification of radio transmission components. Firms must clearly explain in the product packaging or user manual that the product has used e-labeling. Users must be able to access the information without special access codes or an approval process and must be able to access the label within three steps. E-labeling info must be protected so that it can’t be changed or removed. In November 2020, the Korean Radio Research Agency (RRA) issued notice 2020-7 to update some aspects regarding the conformity assessment, including allowing QR code labeling (instead of standard physical labels or e-labels).[69]
Taiwan
Since 2017, Taiwan has allowed firms to present the Regulator National Communications Commission (NCC) label electronically, though the NCC logo must still be physically printed or attached to individual product packaging. Starting in 2016, the Taiwanese Food and Drug Administration (TFDA) published an app that can scan bar codes on drug cartons to access official drug information on the drug authority’s website.[70]
Thailand
Following this, in 2020, Thailand’s Industrial Standards Institute (TISI) at the Ministry of Industry released updated regulations that outline how firms must ensure a standard mark shall be displayed in a manner clearly visible, prominently, and indelible on industrial products or also on their packaging, bundles, wrappers, or parcels. Thailand introduced the new regulation to prevent deceptive practices and protect consumers.[71] It allows manufacturers or the importer of TISI-certified products to affix electronic information by QR code together with the TISI mark. It allows firms to use a code showing the name and location of the factory. The regulation makes electronic labeling mandatory for Thai Industrial Standard-certified products. The new requirement went into effect in January 2021.[72]
United Arab Emirates
In 2020, the Telecommunication Regulatory Authority (TRA) of the United Arab Emirates (UAE) advised of a new label requirement applicable to TRA-approved products. The UAE’s conformity mark must be placed on the equipment box of approved products as well as featured under the display platform in retail stores (or any form of display, such as on websites). E-labelling is allowed, but this does not negate the need to place the mark on the equipment box. The QR code should contain the basic information, as shown in the middle box of photo 5.[73]
Figure 5: The United Arab Emirates’ use of physical label and QR code[74]

United Kingdom
The United Kingdom does not currently allow e-labels for regulatory compliance purposes. The Independent Group for Growth and Regulatory Reform recommended that the country adopt e-labeling, but the government has not endorsed it. The U.K. consumer IoT code of conduct has a certification proposal, with the top three security requirements being mandatory. The United Kingdom is considering labeling requirements but has not yet announced details. It seems like it’ll be a physical label, as the United Kingdom has said in the past that e-labels are not permitted in place of traditional physical labels.
United States
The United States has been a leader in successful e-labeling implementation. According to one study’s estimates, at least 87 percent of the smartphones sold in 2016 and 82 percent of the personal computers sold in 2015 displayed compliance information through e-labeling.[75]
Manufacturers of software-defined radios have been allowed to use e-labels since 2001. The FCC allowed manufacturers of software-defined radios (SDRs) to voluntarily use e-labeling. SDRs are microdevices that lack physical space to get the FCC’s surface label affixed. If a modular transmitter uses an electronic display, the FCC certification must be readily available and visible on the device itself or the device in which it is stored. The user manual must also include instructions on how to access the electronic display, and a copy of these instructions must be included in the application for equipment authorization.
In 2014, the use of e-labeling expanded to devices with an integral or connected display, which covers smartphones, e-readers, notebook computers, all-in-one desktop computers, tablet computers, and Internet radios with a digital display. Products without an integrated display but that still qualify to use e-labeling include products that display content via direct connection to a television, monitor, or similar devices (e.g., set-top boxes, game consoles, media streaming products, desktop personal computers) and those that are required to work in conjunction with another product that has a display (e.g., smartphone, laptop computer, desktop computer, baby monitor, or dedicated companion unit).
That same year, the U.S. Congress passed the Enhance Labeling, Accessing, and Branding of Electronic Licenses Act (E-LABEL act), which directed the FCC to allow manufactures of electronic devices with a screen to display information required by the agency digitally—on the device’s screen—rather than on a label affixed to the device.[76] The FCC is responsible for the certification and labeling of RF devices, in order to verify compliance with the commission’s interference rules. FCC Commissioner O’Rielly outlined how the E-LABEL Act provides a number of benefits, including conveying more information to consumers than is conveyed through regular labels, while not undermining the FCC’s certification process.[77] Most recently, in July 2017, the FCC released new orders that codify and expand the guidance it had previously issued on allowing e-labeling.[78] The Congressional Budget Office estimated that implementing the E-LABEL Act had a negligible effect on net discretionary costs for the FCC.[79]
FCC rules hold that all devices with an integral (nonremovable) screen can now display an e-label digitally on that screen, and up to three steps deep into the device menu.[80] For example, starting from the main or home menu:
▪ Step 1 – accessing submenu (for example, “Settings”)
▪ Step 2 – access submenu (for example, “Legal and Regulatory”)
▪ Step 3 – access submenu (for example, “Regulatory,” wherein applicable regulatory information is displayed)
The user manual must include information on accessing that FCC info, or it can be on a product’s website. Users must be able to access the information without any special access codes or permissions, it must not require any special accessories or plug-ins (e.g., a SIM card), and users must be provided with specific instructions on how to access the information.[81] The information must be secured and implemented in a factory-set-unalterable format.[82]
Relatedly, the U.S. Food and Drug Administration (FDA) is pioneering the concept of Internet-based product information for medical devices and their unique device identification (UDI) number. In 2008, Congress passed legislation calling for a unique device identifier system. The FDA now requires manufacturers, and in some cases distributors, to label each medical device or its packaging with a unique number that can be read by humans and machines. By inputting that number or scanning a barcode, providers should be able to get data about the product from the FDA’s Global UDI Database, such as its serial number, lot number, and manufacture and expiration dates. This builds on far-sighted rules introduced in 2002 through the Medical Device User Fee and Modernization Act (MDUFA), which allowed e-labels to provide all labeling information for prescription devices used in health facilities, but also allowed for health care facilities to still require labeling in paper form.[83]
Vietnam
Vietnam has, as of yet, not implemented e-labeling—but it is slowly developing. A 2017 report submitted by Vietnam to APEC outlining the benefits of and legal framework for e-labeling shows promise that it may be allowed soon.[84]
Vietnam has considered reforms to allow e-labeling as part of broader efforts to create a National Single Window Mechanism and ASEAN Single Window Mechanism and reform the specialized inspection of imported and exported goods and facilitate trade. These draft changes touch on the Law on Organisation of the Government, Law on Product and Goods Quality, and the Law on Measurement. Vietnam’s effort involves both the Minister of Science and Technology and the Department of Product Quality Management at the Ministry of Industry. In 2020, Vietnam’s Ministry of Science and Technology put out guidance on e-labeling under the “Draft decree amending Decree 43 and Draft Circular on presentation of certain compulsory label contents by electronic means.”
The consideration and development of e-labeling rules in Vietnam is indicative of the challenge firms potentially face in dealing with multiple government agencies on e-labeling to ensure a single, clear, and consistent approach to e-labels.
Endnotes
[1]DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU” (Industry paper, June, 2018), https://www.digitaleurope.org/resources/cost-benefits-analysis-on-the-introduction-of-an-e-labelling-scheme-in-europe/.
[3]Nigel Cory, “How E-Labels Can Support Trade and Innovation in ICT” (ITIF, September 25, 2017), https://itif.org/publications/2017/09/25/how-e-labels-can-support-trade-and-innovation-ict.
[4]“Are Labels on Mobile Phones Packaging Effective? A Study of Chile and Colombia” (Luxon Consulting Group, November, 2020), https://www.luxonconsultinggroup.com/are-labels-on-mobile-phone-packaging-effective-a-study-of-chile-and-colombia/.
[5]“ITI Position Paper: E-Labeling” (IT Industry Council, May, 2021), https://www.itic.org/dotAsset/33059c59-b2be-4c33-895f-f95ca4e344f2.pdf.
[6]Jyri Turunen, ”Built-in QR reader on Android,” Medium, (Nov 28, 2018), https://medium.com/turunen/built-in-qr-reader-on-android-696e0f38113b; “Mobile Fact Sheet,” Pew Research Center, published April 7, 2021, https://www.pewresearch.org/internet/fact-sheet/mobile/.
[7]Blue Bite, “The State of QR in 2021,” Blue Bite website, June 21, 2021, https://www.bluebite.com/nfc/qr-code-usage-statistics.
[8]Scanova Blog, ”QR Code Statistics 2021: Up-To-Date Numbers On Global QR Code Usage” (Accessed August 19, 2021), https://scanova.io/blog/qr-code-statistics/#site-header.
[9]Mobileiron, ”QR Codes: Consumer Sentiment Survey” (September, 2020), https://www.mobileiron.com/en/qriosity.
[10]“New Electronic Labeling for Medical Devices,” Obelis Group website, https://www.obelis.net/news/new-electronic-labeling-for-medical-devices/.
[11]European Union Agency for Cybersecurity (ENISA), Cloud Services Scheme (ENISA, December 22, 2020), https://www.enisa.europa.eu/publications/eucs-cloud-service-scheme.
[12]“Cybersecurity Labeling for Consumers: Internet of Things (IoT) Devices and Software,” U.S. National Institute for Standards and Technology, https://www.nist.gov/itl/executive-order-improving-nations-cybersecurity/cybersecurity-labeling-consumers-internet-things.
[13]Ibid.
[14]IT Industry Council, “Cybersecurity Labeling: A Guide for Policymakers” (Industry paper, April, 2021), https://www.itic.org/documents/cybersecurity/ITI_CybersecruityLabeling_Final_Apr2021.pdf.
[15]Jeremy Kirk, “Singapore Expands Consumer IoT Labelling,” Dat Breach Today, January 28, 2021, https://www.databreachtoday.com/singapore-expands-consumer-iot-labelling-a-15874.
[16]“Circular Economy and Material Value Chains,” World Economic Forum, https://www.weforum.org/projects/circular-economy.
[17]Cristina Marcos, “House passes 'E-labeling' bill,” The Hill, September 11, 2014, http://thehill.com/blogs/floor-action/house/217448-house-passes-e-labeling-bill.
[18]DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU.”
[19]Ibid.
[20]Ibid.
[21]“ASEAN-Australia Digital Trade,” Standards Australia, https://www.standards.org.au/engagement-events/international/asean-australia-digital-trade.
[23]Elena Astapenko, “Principles of Labelling for Medical Devices and IVD Medical Devices,” IMDRF, March 2, 2019, http://www.imdrf.org/docs/imdrf/final/technical/imdrf-tech-190321-pl-md-ivd.pdf.
[24]“ISO 20417:2021 Medical devices—Information to be supplied by the manufacturer,” ISO, https://www.iso.org/standard/67943.html.
[25]IT Industry Council, “ITI Position Paper: E-Labeling” (Industry paper, May, 2021), https://www.itic.org/dotAsset/33059c59-b2be-4c33-895f-f95ca4e344f2.pdf.
[26]“Argentina Updates Labeling Requirements,” GMA Consulting Group, https://www.gma.trade/news/argentina-updates-labeling-requirements.
[27]“Argentina Resolution 169/2018,” March 28, 2018, https://www.argentina.gob.ar/normativa/nacional/resoluci%C3%B3n-169-2018-308277/actualizacion.
[28]“Radiocommunications Legislation Amendment (Reform and Modernisation) Act 2020,” https://www.legislation.gov.au/Details/C2020A00151.
[29]DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU.”
[32]“Regulatory News,” https://www.ib-lenhardt.de/en/news/all/.
[33]Notice 2014-DRS1003; “Notice 2014-DRS1003,” Canada, https://www.ic.gc.ca/eic/site/ceb-bhst.nsf/eng/tt00099.html.
[34]“USMCA: Chapter 12: Sectoral Annexes,” https://ustr.gov/sites/default/files/files/agreements/FTA/USMCA/Text/12_Sectoral_Annexes.pdf.
[35]“Multiband/5G Provisional Seal,” May 10, 2021, https://multibanda.cl/sello-provisorio-multibanda-5g/.
[36]“Chile: Resolution 22334,” March 8, 2018, https://www.sec.cl/transparencia/docs2018/Resolucion_22334.pdf.
[37]Undersecretary of Telecommunications of Chile, “Informe Anual del Sector de Telecomunicaciones 2019,” https://www.subtel.gob.cl/estudios-y-estadisticas/informes-sectoriales-anuales/.
[38]“CNCA Adopts E-Labelling Allowance for CCC Marks,” United States Information Technology Office (USITO), Accessed August 11, 2021, http://www.usito.org/news/cnca-adopts-e-labeling-allowance-ccc-marks; DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU.”
[39]“Regulatory News,” https://www.ib-lenhardt.de/en/news/all/.
[40]“EU Regulation 207/2012: Electronic Instructions for Use of Medical Devices,” Official Journal of the European Union, March 10, 2012, https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2012:072:0028:0031:en:PDF.
[41]DigitalEurope, “e-labeling for Europe – key facts and figures” (policy paper, June 22, 2018), https://www.digitaleurope.org/resources/e-labelling-for-europe-key-facts-figures/.
[42]Ibid.
[43]DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU.”
[44]NATIONAL COMMUNICATIONS AUTHORITY, ”TYPE APPROVAL GUIDELINES” (Accra: National Communications Authority, May 2015), https://nca.org.gh/assets/Uploads/Type-Approval-Revised-Guidelines-2015-Final-Revised2.pdf; Nigel Cory, ”How E-Labels Can Support Trade and Innovation in ICT” (ITIF, September 2017), https://www2.itif.org/2017-e-label-support-ict.pdf?_ga=2.217048595.484852342.1628536822-50281195.1623114385.
[45]Indian Ministry of Electronics and Information Technology (MeitY), ”Electronic Labelling (e-labelling) Guidelines” (September 13, 2017), https://www.meity.gov.in/writereaddata/files/E%20LABELLING%20_GUIDELINES.pdf.
[46]“What are the requirements of Indonesia SDPPI Approval Label?” DIMULTI website, https://www.dimulti.id/indonesia-sdppi-approval-label/.
[47]DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU.”
[48]Rie Matsui et al., ”Global Electronic Labeling Initiatives: Updates from Japan, Canada, Europe, US, and Asia” (Global Forum, July 2020), https://globalforum.diaglobal.org/issue/july-2020/global-electronic-labeling-initiatives-updates-from-japan-canada-europe-us-and-asia/; Kurihara Sayaka, “Current and planned e-labeling initiatives in Japan” (Asia Pacific Conference of Pharmaceutical Associations, presentation), https://apac-asia.com/images/achievements/pdf/10th/04_Access%20To%20Innovative%20Medicines%20Session%201%20(e-labeling)/02_Current%20and%20planned%20e-labeling%20initiatives%20in%20Japan_Sayaka%20Kurihara_PMDA.pdf.
[49]Ministry of Health Malaysia, “Requirements for Labeling of Medical Devices” (January 2018), https://portal.mda.gov.my/ms/dokumen/guidance-documents/732-requirements-for-labelling-of-medical-devices-revise/file.html.
[50]“Guideline on Certification Mark for Self Labelling (SL) of Certified Communication Products in Malaysia,” World Trade Organization (WTO), January 2015, https://members.wto.org/crnattachments/2015/TBT/MYS/15_1370_00_e.pdf.
[51][Translated] “Current Technical Provisions,” Mexico’s Federal Institute for Telecommunications, http://www.ift.org.mx/industria/politica-regulatoria/disposiciones-tecnicas?page=1; “Mexico: (Officlal Regulation NOM-106-SCFA-2017),” https://www.dof.gob.mx/normasOficiales/6758/seeco12_C/seeco12_C.html.
[52]“USMCA: Chapter 12: Sectoral Annexes,” https://ustr.gov/sites/default/files/files/agreements/FTA/USMCA/Text/12_Sectoral_Annexes.pdf.
[53]“Label your products,” Radio Spectrum Management Compliance Instructions, accessed August 11, 2021, https://www.rsm.govt.nz/business-individuals/supplier-compliance/steps/step-7/.
[54]“Labelling of Medical Devices,” MedSafe, published September 23, 2016, https://www.medsafe.govt.nz/devices/labelling-and-ifu.asp.
[55]“Why we introduced product authentication mark–SON,” The Sun, June 18, 2020, https://www.sunnewsonline.com/why-we-introduced-product-authentication-mark-son/.
[56]“Nigeria SON – Product Authentication Mark Introduction,” https://www.xds-solutions.com/nigeria-son-product-authentication-mark-pam/.
[57]“About Chestny ZNAK,” https://chestnyznak.ru/en/o-chestnom-znake/.
[58]DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU.”
[59]Ibid.
[60]Ibid.
[61]Requirements for Telecommunication Equipment Labels and Advertisements,” October, 2020, https://www.imda.gov.sg/-/media/Imda/Files/Regulations-and-Licensing/Licensing/Telecommunication/Telecommunication-Equipment-Labels-and-Advertisement/TelecomEqupLabel_Adv_IMDA-V3.pdf.
[62]Launched in 2005, the Telecoms Licensing portal is an online portal that allows suppliers and providers to register their telecommunication equipment and apply and pay for licences via the Internet.
[63]Andy Leck and Ren Jun Lim, ”Singapore - Introduction Of Electronic Labelling ("e-labelling") For Therapeutic Products” (Conventus Law, January 31, 2020), https://www.conventuslaw.com/report/singapore-introduction-of-electronic-labelling-e/; ”Electronic labelling (e-labelling) for Therapeutic Products” (open letter, Singapore Health and Sciences Authority (HSA), August 2019), https://www.hsa.gov.sg/docs/default-source/hprg/regulatory-updates/e-labelling-19-aug-2019.pdf.
[64]“Middle East, G-Mark: New Gulf Conformity Tracking System,” UL website, June 19, 2019, https://www.ul.com/news/middle-east-g-mark-new-gulf-conformity-tracking-system.
[65]“Notice 872 of 2013,” Communications Authority of South Africa, https://autodocbox.com/Motorcycles/97849904-Notice-872-of-2013-independent-communications-authority-of-south-africa.html.
[66]“Government Gazette Staatskoerant,” Republic of South Africa, https://archive.opengazettes.org.za/archive/ZA/2013/government-gazette-ZA-vol-578-no-36786-dated-2013-08-26.pdf
[67]Republic of Korea Radio Waves Act, 2015, https://elaw.klri.re.kr/eng_service/lawView.do?hseq=7162&lang=ENG#:~:text=The%20purpose%20of%20this%20Act,development%20of%20radio%20wave%20technology.
[68]Republic of Korea, “Electrical Appliance and Consumer Products Safety Control Act (2016),” https://elaw.klri.re.kr/eng_mobile/viewer.do?hseq=45624&type=sogan&key=13; Nigel Cory, ”How E-Labels Can Support Trade and Innovation in ICT” (ITIF, September 2017), https://www2.itif.org/2017-e-label-support-ict.pdf?_ga=2.217048595.484852342.1628536822-50281195.1623114385.
[69]“Regulatory News,” https://www.ib-lenhardt.de/en/news/all/; “Korea Updates Conformity Assessment Scheme for Telecom Equipment,” GMA Consult Group, https://www.gma.trade/news/korea-updates-conformity-assessment-scheme-for-telecom-equipment?e27cb8f4_page=2.
[70]Priyanka Chaudhary and Vikram Shetty, ”E-Labeling: Change is Underway” (PharmExec, June 2020), https://www.pharmexec.com/view/e-labeling-change-underway; Product Compliance Specialists, ”Regulatory Update-Taiwan” (August 2017), https://www.kiwa.com/4aaea4/globalassets/uk/pcs/regulatory-updates-2017/regulatory_update_taiwan_ncc_02-aug-17.pdf.
[71]Thai Industrial Standards Institute (TISI), ”Ministerial Regulation Prescribing Description, Production and Method of Displaying of Standard Marks on the Industrial Products B.E.2563” (WTO, 2020), https://docs.wto.org/dol2fe/Pages/FE_Search/FE_S_S009-DP.aspx?language=E&HasEnglishRecord=True&HasFrenchRecord=False&HasSpanishRecord=False&CatalogueIdList=265578,265613,265612,265611,265610,265609,265620,265593,257428,265668&CurrentCatalogueIdIndex=9&Full; https://members.wto.org/crnattachments/2020/TBT/THA/20_4781_00_x.pdf.
[72]Umeyama Kenichi, ”Thailand: QR Code required for labels on TISI-certified products” (Enviliance Asia, October 28, 2020), https://enviliance.com/regions/southeast-asia/th/report_1162
[73]“Regulation: Telecommunications Equipment,” Egypt’s Telecommunications Regulatory Authority, October 2, 2018, https://www.tdra.gov.ae/assets/2X256hMb.pdf.aspx.
[74]Ibid.
[75]DigitalEurope and the Mobile & Wireless Forum, “Cost-Benefits analysis on the introduction of an e-labelling scheme in the EU.”
[76]E-LABEL Act of 2014, H.R. 5161, 113th Cong. (2014), https://www.congress.gov/bill/113th-congress/house-bill/5161/text.
[77]Michael O’Rielly, “E-Labeling Deserves Serious Consideration,” Federal Communications Commission blog post, April 25, 2014, https://www.fcc.gov/news-events/blog/2014/04/25/e-labeling-deserves-serious-consideration.
[78]U.S. Federal Communications Commission, First Report and Order (17-93), https://apps.fcc.gov/oetcf/kdb/forms/FTSSearchResultPage.cfm?switch=P&id=27980.
[79]H.R. 5161, Enhance Labeling, Accessing, and Branding of Electronic Licenses Act of 2014, Congressional Budget Office (CBO), (July 30, 2014), https://www.cbo.gov/publication/45666.
[80]Federal Communications Commission, “Electronic Labeling Guidance” (Washington, D.C.: Federal Communications Commission Office of Engineering and Technology Laboratory Division, July, 2014), https://apps.fcc.gov/kdb/GetAttachment.html?id=KvMvDHtHyDtJ4FB3x0mEwA%3D%3D.
[81]Ibid.
[82]Ibid.
[83]“What Medical Device Manufacturers Need to Know About E-Labeling,” QTS website, accessed August 29, 2017, https://qtspackage.com/2016/what-medical-device-manufacturers-need-to-know-about-e-labeling/.
[84]“E-Labeling for ICT Products - A Perspective from Viet Nam,” Asia-Pacific Economic Cooperation (APEC), August 2017, http://mddb.apec.org/Documents/2017/SCSC/WKSP2/17_scsc_wksp2_011.pdf.