
Senate Hearing Inadvertently Shows Why Antitrust Policy Is the Wrong Prescription for What Ails Drug Markets
Four out of five Americans say prescription drug prices are unreasonably high, according to a recent KFF survey—and people across the political spectrum say pharma profits are a major contributing factor. So it came as no surprise earlier this month when Sen. Amy Klobuchar (D-MN) called a hearing in the Senate Subcommittee on Competition Policy, Antitrust, and Consumer Rights to scrutinize alleged anticompetitive conduct in prescription drug markets. But in the end, the hearing merely demonstrated that, to the extent there is a problem with drug prices in America, antitrust policy is the wrong tool to address it.
First, some important context. The truth is that generic drug prices are lower in America than in other countries—57 percent lower than in Canada, 58 percent lower than in France, and 68 percent lower than in the United Kingdom—and generic drugs account for 91 percent of dispensing generic medicine prescriptions. More broadly, the notion that prescription drug prices in the United States are out of control is a case of perception overshadowing reality. For instance, according to the Peterson Center on Healthcare and Kaiser Family Foundation, the share of U.S. healthcare spending going toward retail prescription drugs has been remarkably consistent from 2000 to 2017 and was projected to grow only modestly from 2018 to 2027. And according to the U.S. Bureau of Labor Statistics, U.S. hospital prices increased by more than one-third more than U.S. drug prices, from 1999 to 2000. Drug prices grew only slightly more than prices for doctors (195 percent versus 165 percent) and only moderately more than the increase in the overall U.S. consumer price index over this period (155 percent). Moreover, net per capita spending on prescription medicines has remained effectively flat, increasing just 0.5 percent on average over the past 10 years.
That biopharmaceutical companies have brought numerous new-to-the-world treatment innovations for a variety of ailments such as cancers, cardiovascular disease, and hepatitis C—all while holding medicines’ share of health spending nearly constant—is reflective of the highly competitive structure of the U.S. market. To be sure, it’s important to balance the cost of medicines against the expense of making them and the value they deliver. For instance, the more information technologies and biotechnologies can be deployed to enhance biotech R&D efficiency, the more it can help rein in rapidly rising drug discovery and development costs. Reforming reimbursement systems, such as by capping patients’ coinsurance payments on Part D drug plans and passing on to patients more of the rebates paid by biopharmaceutical companies, also could help reduce their out-of-pocket costs.
That background is important to keep in mind when parsing the claims aired in the Senate subcommittee hearing that Sen. Klobuchar organized. Very few of the issues raised regarding U.S. branded drug prices were antitrust matters.
First, Dr. Rachel Moodie from Fresenius Kabi asserted that “patent thickets”—secondary patents filed after the core drug patents are filed—are the “root cause” of high drug prices. Acknowledging that “not all secondary patents are bad,” Dr. Moodie nevertheless pointed out that some secondary patents may be of “low quality”—meaning, not true innovations but a move to extend the intellectual property rights (IPR) surrounding a drug. Should that ever be true, the U.S. Patent and Trademark Office may revise its approach on current rules to ensure they do not unduly delay the entry of generics. However, the recently passed CREATES Act facilitates the development of generics. Given the lack of sufficient experience amassed since its entry into force in 2020, it’s not yet necessary to refine existing laws. Moreover, it is precisely consistent with President Biden’s executive order, which directs the secretary of Health and Human Services to “support the market entry of lower-cost generic drugs and biosimilars” and to “continue the implementation of the law widely known as the CREATES Act of 2019.”
In other words, improved guidance within the patent system’s current rules may help address the few instances of “patent thickets.” Antitrust rules are highly inappropriate to deal with this IPR-intensive policy as they may cause more harm than good.
Second, the hearing multiple times raised the concern that patients don’t see sufficient discounts and rebates at the drug counter. Another way to ensure that drug prices at the counter better reflect the prices of drugs charged by manufacturers would be to spur competition in other markets of the pharmaceutical supply chain—namely, among wholesalers and pharmacies. Just as President Biden’s executive order would allow the sale of hearing aids over the counter, easing the sale of drugs by multiple actors could help reduce drug prices. Already, disruptors such as Amazon represent the promise of driving drug prices down by spurring competition in the pharmacy market.
But again, this is not an antitrust issue: It is a regulatory issue. Deregulating drug sales at pharmacies’ counters (or at different shops for non-prescription drugs) would help patients benefit from the real prices drug manufacturers charge without costly intermediaries.
Another example of how drug prices are not an antitrust concern came when the committee members suggested importing some drugs from Canada, since drug prices are lower there. To be sure, the prospect of such imports would likely lead to Canada’s ban on export in fear of drug shortages, as this already occurred when the Trump administration issued an executive order to implement the idea. Moreover, importing Canada’s drug price control system would thwart U.S. pharmaceutical innovation capabilities by undermining drug manufacturers to strengthen and develop their research capabilities for new drugs. Finally, it would raise trade issues of parallel import at the World Trade Organization, as other countries may complain not to be offered similar privileges given their similar safety standards.
In other words, the idea of importing drugs from Canada, reiterated in President Biden’s executive order, is a trade issue—not an antitrust issue.
In fact, the sole antitrust concern that relates to drug prices pares down to the price opacity of pharmacy benefit managers (PBMs). The lack of price transparency enables PBMs to avoid passing on to consumers the rebates and discounts drug manufacturers offer because of PBMs’ buying power. The FTC may consider ways to foster price transparency among PBMs so that patients can benefit from the net prices charged by drug manufacturers to PBMs (meaning prices minus rebates). This was made clear in a 2019 congressional hearing, especially concerning insulin prices. The discrepancy between list price charged by drug manufacturers and net prices proves particularly blatant, as Hernandez et al. pointed out in a JAMA article last year:
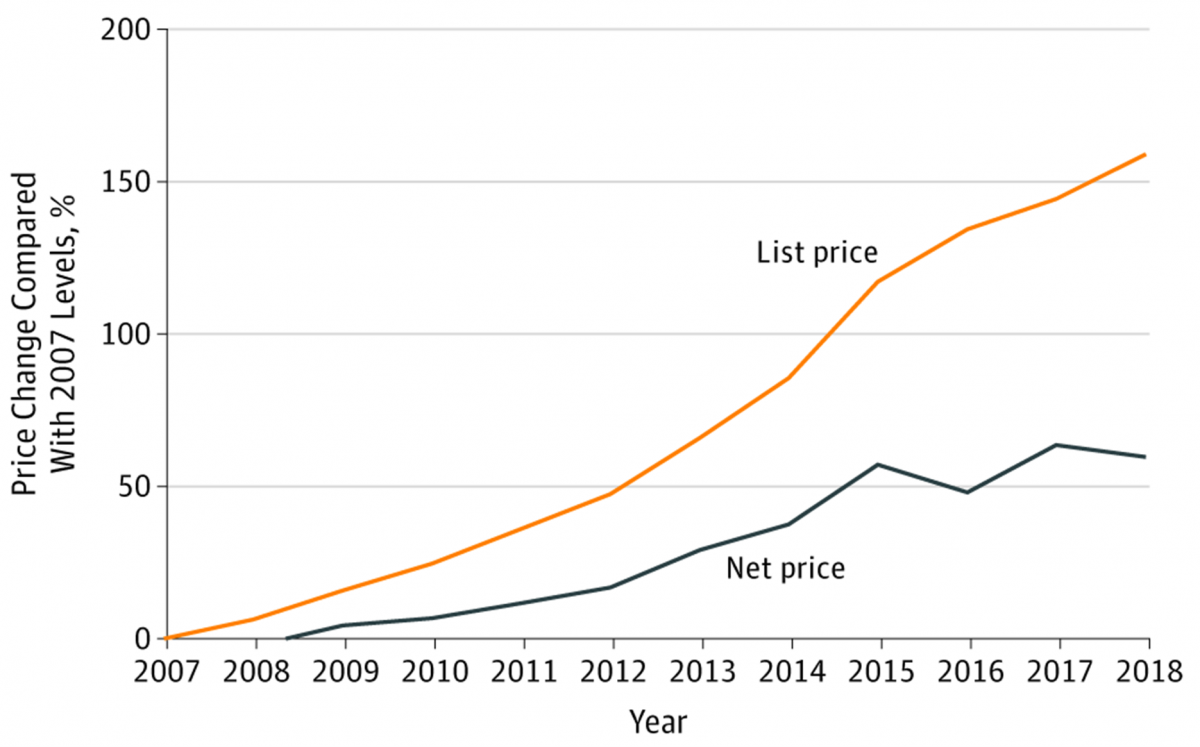
This discrepancy is coined the “gross-to-net-bubble”: Drug prices increase whereas drug manufacturers earn less from drugs. This decrease in drug revenues hampers drug discoveries.
For patients to see a decrease in drug prices at the counter, PBMs need to pass on the huge rebates and discounts they secure from drug manufacturers. Enhanced price transparency by PBMs would reduce drug prices for patients without stifling innovation.
The leadership of the U.S. pharmaceutical industry in drug innovation remains unparalleled. We should keep it that way and avoid common misconceptions. Expanding the reach of antitrust to tackle pro-innovative and pro-competitive conduct by drug manufacturers while overlooking paths for reforms such as enhancing PBMs’ price transparency, increasing competition among pharmacies and wholesalers, and clarifying the CREATES Act could create the worst of both worlds—no drug price reduction and deterred innovation.
The recent Senate subcommittee hearing failed to acknowledge the limits of antitrust and identify concrete steps to pursue these reasonable paths for reforms.

